Identification and Characterization of the 'Missing' Terminal Enzyme for Siroheme Biosynthesis in Alpha-Proteobacteria.
Bali, S., Rollauer, S., Roversi, P., Raux-Deery, E., Lea, S.M., Warren, M.J., Ferguson, S.J.(2014) Mol Microbiol 92: 153
- PubMed: 24673795 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1111/mmi.12542
- Primary Citation Related Structures:
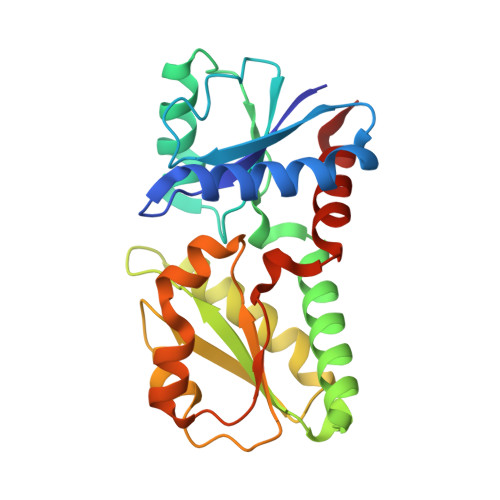
4CCS - PubMed Abstract:
It has recently been shown that the biosynthetic route for both the d1 -haem cofactor of dissimilatory cd1 nitrite reductases and haem, via the novel alternative-haem-synthesis pathway, involves siroheme as an intermediate, which was previously thought to occur only as a cofactor in assimilatory sulphite/nitrite reductases. In many denitrifiers (which require d1 -haem), the pathway to make siroheme remained to be identified. Here we identify and characterize a sirohydrochlorin-ferrochelatase from Paracoccus pantotrophus that catalyses the last step of siroheme synthesis. It is encoded by a gene annotated as cbiX that was previously assumed to be encoding a cobaltochelatase, acting on sirohydrochlorin. Expressing this chelatase from a plasmid restored the wild-type phenotype of an Escherichia coli mutant-strain lacking sirohydrochlorin-ferrochelatase activity, showing that this chelatase can act in the in vivo siroheme synthesis. A ΔcbiX mutant in P. denitrificans was unable to respire anaerobically on nitrate, proving the role of siroheme as a precursor to another cofactor. We report the 1.9 Å crystal structure of this ferrochelatase. In vivo analysis of single amino acid variants of this chelatase suggests that two histidines, His127 and His187, are essential for siroheme synthesis. This CbiX can generally be identified in α-proteobacteria as the terminal enzyme of siroheme biosynthesis.
- Department of Biochemistry, University of Oxford, South Parks Road, Oxford, OX1 3QU, UK.
Organizational Affiliation: