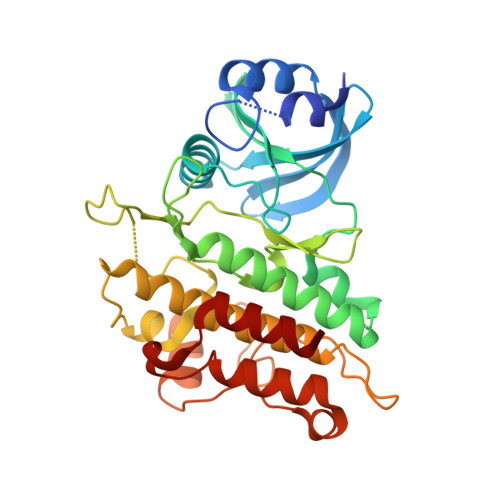
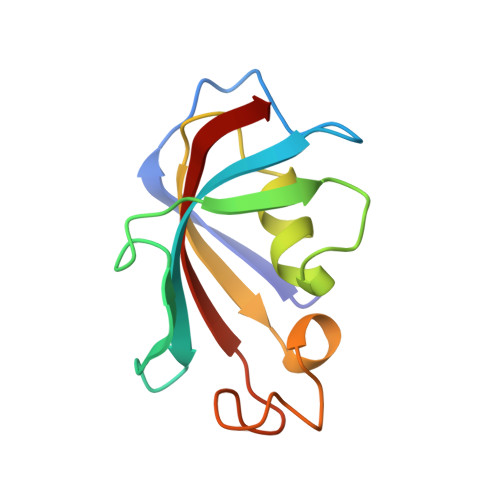
Crystal Structure of Human Acvr1 (Alk2) in Complex with Fkbp12.6 And Dorsomorphin
Williams, E., Riesebos, E., Vollmar, M., Krojer, T., Bradley, A., Shrestha, L., Kupinska, K., von Delft, F., Arrowsmith, C.H., Edwards, A.M., Bountra, C., Bullock, A.(null) Ph D Thesis