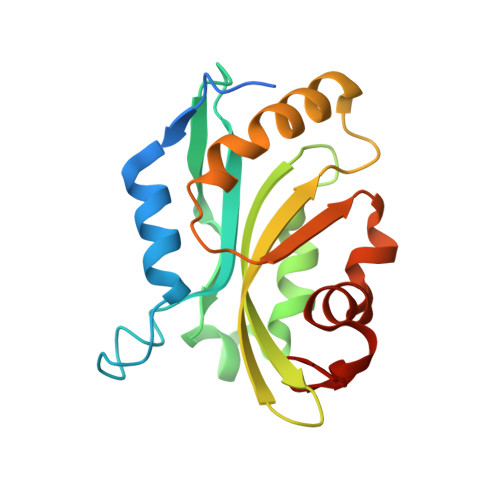
Analysis of the Human Cofilin1 Structure Reveals Conformational Changes Required for Actin-Binding
Klejnot, M., Gabrielsen, M., Cameron, J., Mleczak, A., Talapatra, S.K., Kozielski, F., Pannifer, A., Olson, M.F.(2013) Acta Crystallogr D Biol Crystallogr 69: 1780
- PubMed: 23999301 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S0907444913014418
- Primary Citation Related Structures:
4BEX - PubMed Abstract:
The actin cytoskeleton is the chassis that gives a cell its shape and structure, and supplies the power for numerous dynamic processes including motility, endocytosis, intracellular transport and division. To perform these activities, the cytoskeleton undergoes constant remodelling and reorganization. One of the major actin-remodelling families are the cofilin proteins, made up of cofilin 1, cofilin 2 and actin-depolymerizing factor (ADF), which sever aged ADP-associated actin filaments to reduce filament length and provide new potential nucleation sites. Despite the significant interest in cofilin as a central node in actin-cytoskeleton dynamics, to date the only forms of cofilin for which crystal structures have been solved are from the yeast, Chromalveolata and plant kingdoms; none have previously been reported for an animal cofilin protein. Two distinct regions in animal cofilin are significantly larger than in the forms previously crystallized, suggesting that they would be uniquely organized. Therefore, it was sought to determine the structure of human cofilin 1 by X-ray crystallography to elucidate how it could interact with and regulate dynamic actin-cytoskeletal structures. Although wild-type human cofilin 1 proved to be recalcitrant, a C147A point mutant yielded crystals that diffracted to 2.8 Å resolution. These studies revealed how the actin-binding helix undergoes a conformational change that increases the number of potential hydrogen bonds available for substrate binding.
- Beatson Institute for Cancer Research, Garscube Estate, Switchback Road, Glasgow G61 1BD, Scotland.
Organizational Affiliation: