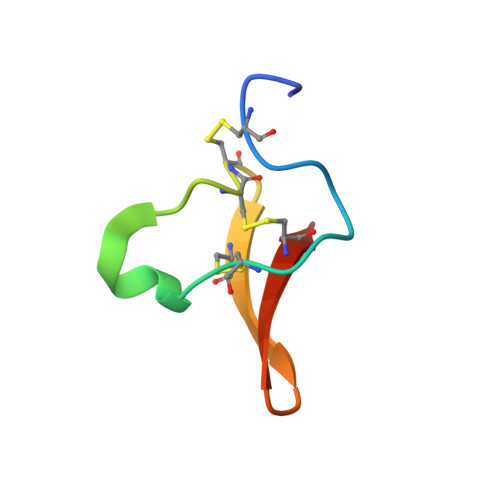
Solution Structures of Two Homologous Venom Peptides from Sicarius Dolichocephalus.
Loening, N.M., Wilson, Z.N., Zobel-Thropp, P.A., Binford, G.J.(2013) PLoS One 8: 54401
- PubMed: 23342149 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0054401
- Primary Citation Related Structures:
4B2U, 4B2V - PubMed Abstract:
We present solution-state NMR structures for two putative venom peptides from Sicarius dolichocephalus. These peptides were identified from cDNA libraries created from venom gland mRNA and then recombinantly expressed. They are the first structures from any species of Sicarius spiders, and the first peptide structures for any haplogyne spiders. These peptides are homologous to one another, and while they have at most only 20% sequence identity with known venom peptides their structures follow the inhibitor cystine knot motif that has been found in a broad range of venom peptides.
- Chemistry Department, Lewis & Clark College, Portland, Oregon, United States of America. loening@lclark.edu
Organizational Affiliation: