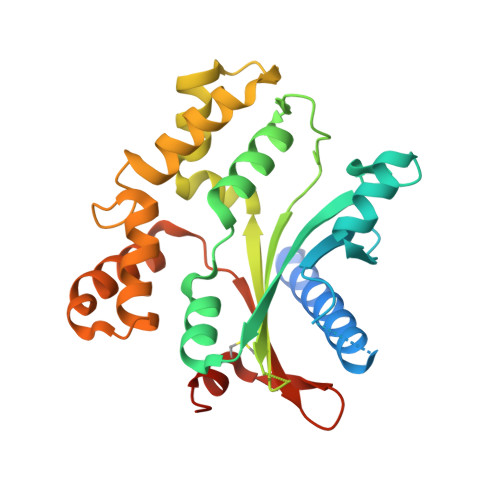
Crystal Structure of Atv(Orf273), a New Fold for a Thermo-and Acido-Stable Protein from the Acidianus Two-Tailed Virus.
Felisberto-Rodrigues, C., Blangy, S., Goulet, A., Vestergaard, G., Cambillau, C., Garrett, R.A., Ortiz-Lombardia, M.(2012) PLoS One 7: 45847
- PubMed: 23056221 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0045847
- Primary Citation Related Structures:
4ART, 4ATS - PubMed Abstract:
Acidianus two-tailed virus (ATV) infects crenarchaea of the genus Acidianus living in terrestrial thermal springs at extremely high temperatures and low pH. ATV is a member of the Bicaudaviridae virus family and undergoes extra-cellular development of two tails, a process that is unique in the viral world. To understand this intriguing phenomenon, we have undertaken structural studies of ATV virion proteins and here we present the crystal structure of one of these proteins, ATV(ORF273). ATV(ORF273) forms tetramers in solution and a molecular envelope is provided for the tetramer, computed from small-angle X-ray scattering (SAXS) data. The crystal structure has properties typical of hyperthermostable proteins, including a relatively high number of salt bridges. However, the protein also exhibits flexible loops and surface pockets. Remarkably, ATV(ORF273) displays a new α + β protein fold, consistent with the absence of homologues of this protein in public sequence databases.
- CNRS, Aix-Marseille Université, AFMB, UMR 7257, Campus de Luminy, Marseille, France.
Organizational Affiliation: