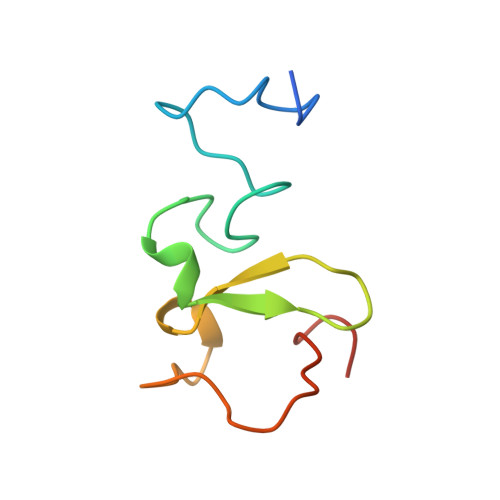
The Phd Finger of Human Uhrf1 Reveals a New Subgroup of Unmethylated Histone H3 Tail Readers.
Lallous, N., Legrand, P., Mcewen, A.G., Ramon-Maiques, S., Samama, J.P., Birck, C.(2011) PLoS One 6: 27599
- PubMed: 22096602 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0027599
- Primary Citation Related Structures:
3ZVY, 3ZVZ - PubMed Abstract:
The human UHRF1 protein (ubiquitin-like containing PHD and RING finger domains 1) has emerged as a potential cancer target due to its implication in cell cycle regulation, maintenance of DNA methylation after replication and heterochromatin formation. UHRF1 functions as an adaptor protein that binds to histones and recruits histone modifying enzymes, like HDAC1 or G9a, which exert their action on chromatin. In this work, we show the binding specificity of the PHD finger of human UHRF1 (huUHRF1-PHD) towards unmodified histone H3 N-terminal tail using native gel electrophoresis and isothermal titration calorimetry. We report the molecular basis of this interaction by determining the crystal structure of huUHRF1-PHD in complex with the histone H3 N-terminal tail. The structure reveals a new mode of histone recognition involving an extra conserved zinc finger preceding the conventional PHD finger region. This additional zinc finger forms part of a large surface cavity that accommodates the side chain of the histone H3 lysine K4 (H3K4) regardless of its methylation state. Mutation of Q330, which specifically interacts with H3K4, to alanine has no effect on the binding, suggesting a loose interaction between huUHRF1-PHD and H3K4. On the other hand, the recognition appears to rely on histone H3R2, which fits snugly into a groove on the protein and makes tight interactions with the conserved aspartates D334 and D337. Indeed, a mutation of the former aspartate disrupts the formation of the complex, while mutating the latter decreases the binding affinity nine-fold.
- Institut National de Santé et de Recherche Médicale U964/Centre National de Recherche Scientifique UMR 7104/Université de Strasbourg, Illkirch, France. nlallous@cnio.es
Organizational Affiliation: