Catalytic Function and Substrate Recognition of the Pectate Lyase from Thermotoga Maritima
McDonough, M.A., Thymark, M., Frisner, H., Hotchkiss, A., Sonksen, C., Bjornvad, M., Johansen, K.S., Larsen, S.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| PECTATE TRISACCHARIDE-LYASE | 340 | Thermotoga maritima | Mutation(s): 3 EC: 4.2.2.22 | 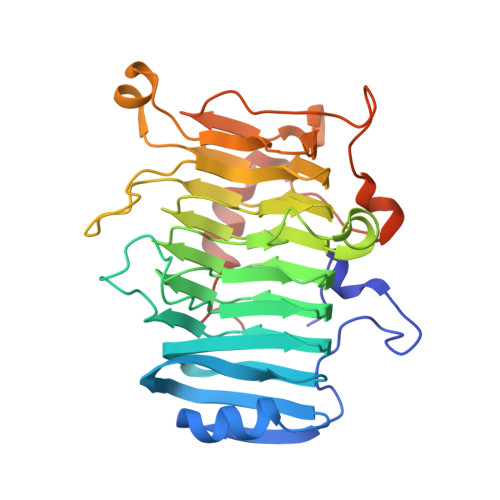 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9WYR4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PO4 Download:Ideal Coordinates CCD File | E [auth A] | PHOSPHATE ION O4 P NBIIXXVUZAFLBC-UHFFFAOYSA-K |  | ||
| GOL Download:Ideal Coordinates CCD File | C [auth A], D [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 80.602 | α = 90 |
| b = 80.602 | β = 90 |
| c = 148.819 | γ = 120 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| DENZO | data reduction |
| EPMR | phasing |