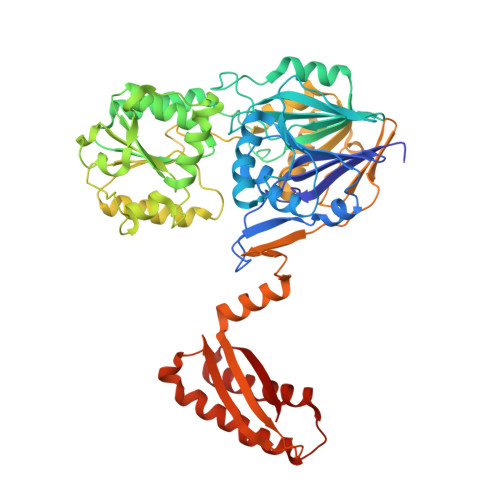
Unusual, Dual Endo- and Exonuclease Activity in the Degradosome Explained by Crystal Structure Analysis of Rnase J1.
Newman, J.A., Hewitt, L., Rodrigues, C., Solovyova, A., Harwood, C.R., Lewis, R.J.(2011) Structure 19: 1241
- PubMed: 21893285
- DOI: https://doi.org/10.1016/j.str.2011.06.017
- Primary Citation Related Structures:
3ZQ4 - PubMed Abstract:
RNase J is an essential enzyme in Bacillus subtilis with unusual dual endonuclease and 5'-to-3' exonuclease activities that play an important role in the maturation and degradation of mRNA. RNase J is also a component of the recently identified "degradosome" of B. subtilis. We report the crystal structure of RNase J1 from B. subtilis to 3.0 Å resolution, analysis of which reveals it to be in an open conformation suitable for binding substrate RNA. RNase J is a member of the β-CASP family of zinc-dependent metallo-β-lactamases. We have exploited this similarity in constructing a model for an RNase J1:RNA complex. Analysis of this model reveals candidate-stacking interactions with conserved aromatic side chains, providing a molecular basis for the observed enzyme activity. Comparisons of the B. subtilis RNase J structure with related enzymes reveal key differences that provide insights into conformational changes during catalysis and the role of the C-terminal domain.
- Institute for Cell and Molecular Biosciences, Newcastle University, Newcastle-upon-Tyne NE2 4HH, UK.
Organizational Affiliation: