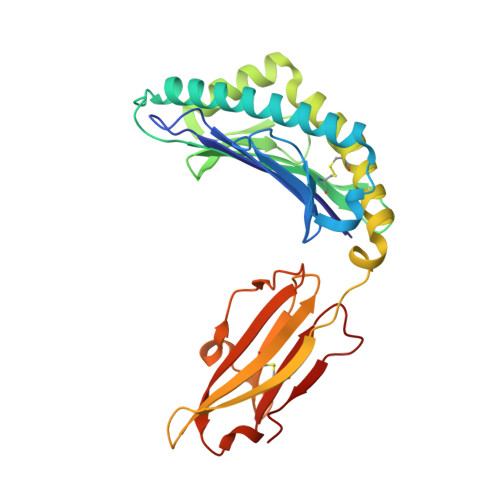
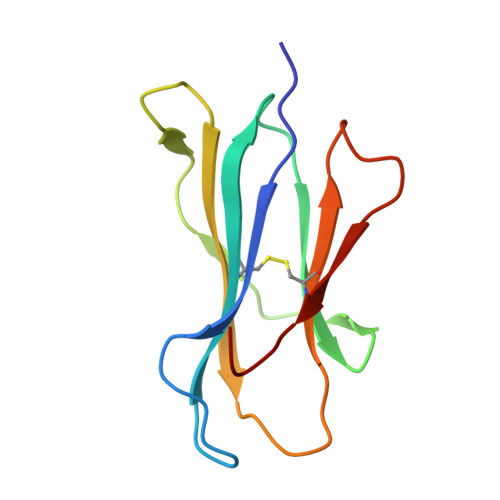
The interaction of KIR3DL1*001 with HLA class I molecules is dependent upon molecular microarchitecture within the Bw4 epitope
Saunders, P.M., Vivian, J.P., Baschuk, N., Beddoe, T., Widjaja, J., O'Connor, G.M., Hitchen, C., Pymm, P., Andrews, D.M., Gras, S., McVicar, D.W., Rossjohn, J., Brooks, A.G.(2015) J Immunol 194: 781-789
- PubMed: 25480565 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.4049/jimmunol.1402542
- Primary Citation Related Structures:
3X11, 3X12, 3X13, 3X14 - PubMed Abstract:
The killer cell Ig-like receptor 3DL1 (KIR3DL1) inhibits activation of NK cells upon interaction with HLA class I molecules such as HLA-B*57:01, which contains the Bw4 epitope spanning residues 77-83 (e.g., NLRIALR), and not with HLA allomorphs that possess the Bw6 motif (e.g., HLA-B*08:01), which differ at residues 77, 80, 81, 82, and 83. Although Bw4 residues Ile(80) and Arg(83) directly interact with KIR3DL1*001, their precise role in determining KIR3DL1-HLA-Bw4 specificity remains unclear. Recognition of HLA-B*57:01 by either KIR3DL1(+) NK cells or the NK cell line YTS transfected with KIR3DL1*001 was impaired by mutation of residues 80 and 83 of HLA-B*57:01 to the corresponding amino acids within the Bw6 motif. Conversely, the simultaneous introduction of three Bw4 residues at positions 80, 82, and 83 into HLA-B*08:01 conferred an interaction with KIR3DL1*001. Structural analysis of HLA-B*57:01, HLA-B*08:01, and mutants of each bearing substitutions at positions 80 and 83 revealed that Ile(80) and Arg(83) within the Bw4 motif constrain the conformation of Glu(76), primarily through a salt bridge between Arg(83) and Glu(76). This salt bridge was absent in HLA-Bw6 molecules as well as position 83 mutants of HLA-B*57:01. Mutation of the Bw4 residue Ile(80) also disrupted this salt bridge, providing further insight into the role that position 80 plays in mediating KIR3DL1 recognition. Thus, the strict conformation of HLA-Bw4 allotypes, held in place by the Glu(76)-Arg(83) interaction, facilitates KIR3DL1 binding, whereas Bw6 allotypes present a platform on the α1 helix that is less permissive for KIR3DL1 binding.
- Department of Microbiology and Immunology, Peter Doherty Institute for Infection and Immunity, The University of Melbourne, Parkville, Victoria 3010, Australia.
Organizational Affiliation: