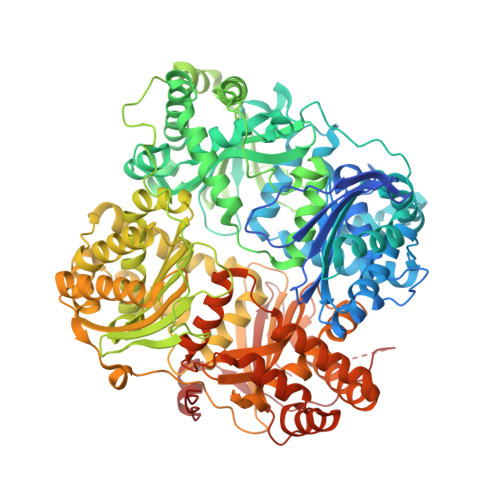
Anion Activation Site of Insulin-degrading Enzyme.
Noinaj, N., Song, E.S., Bhasin, S., Alper, B.J., Schmidt, W.K., Hersh, L.B., Rodgers, D.W.(2012) J Biological Chem 287: 48-57
- PubMed: 22049080 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M111.264614
- Primary Citation Related Structures:
3TUV - PubMed Abstract:
Insulin-degrading enzyme (IDE) (insulysin) is a zinc metallopeptidase that metabolizes several bioactive peptides, including insulin and the amyloid β peptide. IDE is an unusual metallopeptidase in that it is allosterically activated by both small peptides and anions, such as ATP. Here, we report that the ATP-binding site is located on a portion of the substrate binding chamber wall arising largely from domain 4 of the four-domain IDE. Two variants having residues in this site mutated, IDEK898A,K899A,S901A and IDER429S, both show greatly decreased activation by the polyphosphate anions ATP and PPPi. IDEK898A,K899A,S901A is also deficient in activation by small peptides, suggesting a possible mechanistic link between the two types of allosteric activation. Sodium chloride at high concentrations can also activate IDE. There are no observable differences in average conformation between the IDE-ATP complex and unliganded IDE, but regions of the active site and C-terminal domain do show increased crystallographic thermal factors in the complex, suggesting an effect on dynamics. Activation by ATP is shown to be independent of the ATP hydrolysis activity reported for the enzyme. We also report that IDEK898A,K899A,S901A has reduced intracellular function relative to unmodified IDE, consistent with a possible role for anion activation of IDE activity in vivo. Together, the data suggest a model in which the binding of anions activates by reducing the electrostatic attraction between the two halves of the enzyme, shifting the partitioning between open and closed conformations of IDE toward the open form.
- Department of Molecular and Cellular Biochemistry and the Center for Structural Biology, University of Kentucky, Lexington, Kentucky 40536.
Organizational Affiliation: