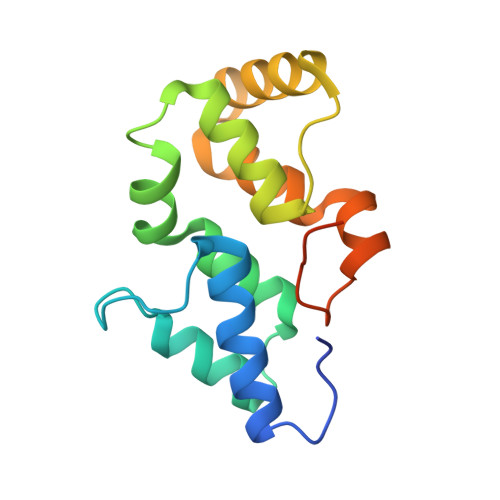
Crystal structure of the N-terminal domain of linker L(R) and the assembly of cyanobacterial phycobilisome rods
Gao, X., Zhang, N., Wei, T.-D., Su, H.-N., Xie, B.-B., Dong, C.-C., Zhang, X.-Y., Chen, X.-L., Zhou, B.-C., Wang, Z.-X., Wu, J.-W., Zhang, Y.-Z.(2011) Mol Microbiol 82: 698-705
- PubMed: 21923764 Search on PubMed
- DOI: https://doi.org/10.1111/j.1365-2958.2011.07844.x
- Primary Citation Related Structures:
3NPH - PubMed Abstract:
Phycobilisomes are light-harvesting supramolecular complexes in cyanobacteria and red algae. Linkers play a pivotal role in the assembly and energy transfer modulation of phycobilisomes. However, how linkers function remains unclear due to the lack of structural and biochemical studies of linkers, especially the N-terminal domain of L(R) (pfam00427). Here, we report the crystal structure of the pfam00427 domain of the linker L(R) (30) from Synechocystis sp. PCC 6803 at 1.9 Å. The pfam00427 presents as a previously uncharacterized point symmetric six α-helix bundle. To elucidate the binding style of pfam00427 in the C-phycocyanin (C-PC) (αβ)(6) hexamer, we fixed pfam00427 computationally into the C-PC (αβ)(6) inner cavity using the program AutoDock. Combined with a conserved 'C-PC binding patch' on pfam00427 identified, we arrived at a model for the pfam00427-C-PC (αβ)(6) complex. This model was further optimized and evaluated as a reasonable result by a molecular dynamics simulation. In the resulting model, the pfam00427 domain is stably positioned in the central hole of the C-PC trimer. Moreover, the L(RT) (pfam01383) was docked into our pfam00427-C-PC model to generate a complete phycobilisome rod in which the linkers join individual biliprotein hexamers.
- State Key Laboratory of Microbial Technology, Marine Biotechnology Research Center, Shandong University, Jinan 250100, China.
Organizational Affiliation: