Structural investigation and inhibitor studies on Carbonic Anhydrase II
Schulze Wischeler, J., Sandner, N.U., Haake, M., Supuran, C., Heine, A., Klebe, G.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Carbonic anhydrase 2 | 265 | Homo sapiens | Mutation(s): 0 Gene Names: CA2 EC: 4.2.1.1 (PDB Primary Data), 4.2.1.69 (UniProt) | 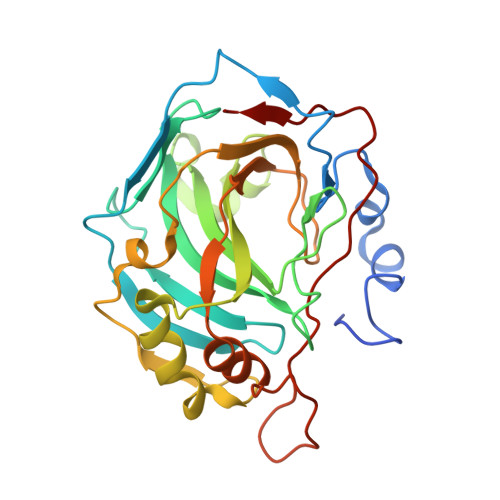 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P00918 GTEx: ENSG00000104267 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00918 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| BE7 Download:Ideal Coordinates CCD File | D [auth A] | (4-CARBOXYPHENYL)(CHLORO)MERCURY C7 H5 Cl Hg O2 YFZOUMNUDGGHIW-UHFFFAOYSA-M |  | ||
| BE9 Download:Ideal Coordinates CCD File | C [auth A] | phenyl N-sulfamoylmorpholine-4-carboximidoate C11 H15 N3 O4 S ODXJJHKPSWXDMI-QBFSEMIESA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | B [auth A] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 42.2 | α = 90 |
| b = 41.9 | β = 104.2 |
| c = 72.3 | γ = 90 |
| Software Name | Purpose |
|---|---|
| MAR345 | data collection |
| PHASER | phasing |
| SHELXL-97 | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| SHELX | phasing |