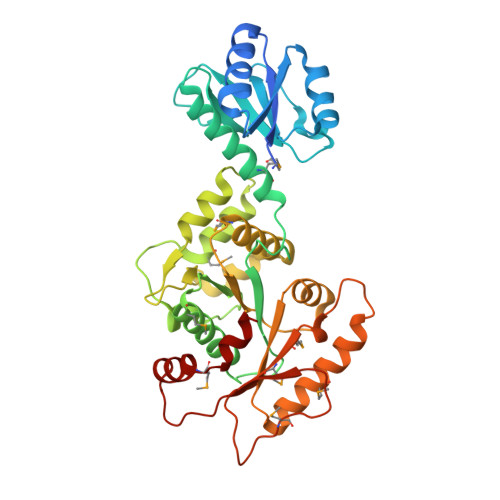
Structure of the pilus assembly protein TadZ from Eubacterium rectale: implications for polar localization.
Xu, Q., Christen, B., Chiu, H.J., Jaroszewski, L., Klock, H.E., Knuth, M.W., Miller, M.D., Elsliger, M.A., Deacon, A.M., Godzik, A., Lesley, S.A., Figurski, D.H., Shapiro, L., Wilson, I.A.(2012) Mol Microbiol 83: 712-727
- PubMed: 22211578 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1111/j.1365-2958.2011.07954.x
- Primary Citation Related Structures:
3FKQ - PubMed Abstract:
The tad (tight adherence) locus encodes a protein translocation system that produces a novel variant of type IV pili. The pilus assembly protein TadZ (called CpaE in Caulobacter crescentus) is ubiquitous in tad loci, but is absent in other type IV pilus biogenesis systems. The crystal structure of TadZ from Eubacterium rectale (ErTadZ), in complex with ATP and Mg(2+) , was determined to 2.1 Å resolution. ErTadZ contains an atypical ATPase domain with a variant of a deviant Walker-A motif that retains ATP binding capacity while displaying only low intrinsic ATPase activity. The bound ATP plays an important role in dimerization of ErTadZ. The N-terminal atypical receiver domain resembles the canonical receiver domain of response regulators, but has a degenerate, stripped-down 'active site'. Homology modelling of the N-terminal atypical receiver domain of CpaE indicates that it has a conserved protein-protein binding surface similar to that of the polar localization module of the social mobility protein FrzS, suggesting a similar function. Our structural results also suggest that TadZ localizes to the pole through the atypical receiver domain during an early stage of pili biogenesis, and functions as a hub for recruiting other pili components, thus providing insights into the Tad pilus assembly process.
- Joint Center for Structural Genomics.
Organizational Affiliation: