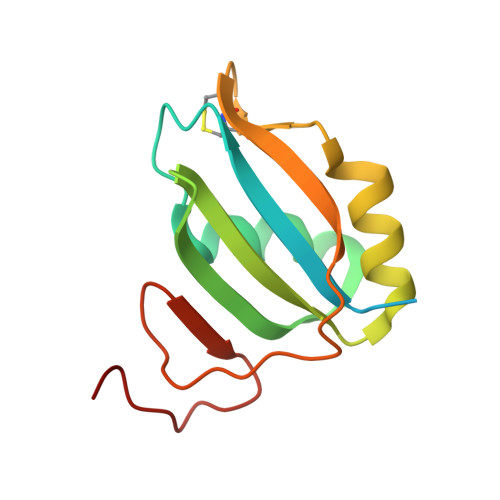
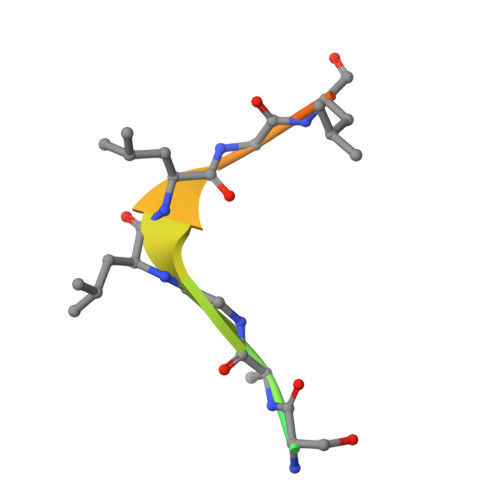
Crystallographic Analysis of Polypyrimidine Tract-Binding Protein-Raver1 Interactions Involved in Regulation of Alternative Splicing.
Joshi, A., Coelho, M.B., Kotik-Kogan, O., Simpson, P.J., Matthews, S.J., Smith, C.W., Curry, S.(2011) Structure 19: 1816
- PubMed: 22153504 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2011.09.020
- Primary Citation Related Structures:
3ZZY, 3ZZZ - PubMed Abstract:
The polypyrimidine tract-binding protein (PTB) is an important regulator of alternative splicing. PTB-regulated splicing of α-tropomyosin is enhanced by Raver1, a protein with four PTB-Raver1 interacting motifs (PRIs) that bind to the helical face of the second RNA recognition motif (RRM2) in PTB. We present the crystal structures of RRM2 in complex with PRI3 and PRI4 from Raver1, which--along with structure-based mutagenesis--reveal the molecular basis of their differential binding. High-affinity binding by Raver1 PRI3 involves shape-matched apolar contacts complemented by specific hydrogen bonds, a new variant of an established mode of peptide-RRM interaction. Our results refine the sequence of the PRI motif and place important structural constraints on functional models of PTB-Raver1 interactions. Our analysis indicates that the observed Raver1-PTB interaction is a general mode of binding that applies to Raver1 complexes with PTB paralogues such as nPTB and to complexes of Raver2 with PTB.
- Division of Cell and Molecular Biology, Imperial College, Exhibition Road, London SW7 2AZ, UK.
Organizational Affiliation: