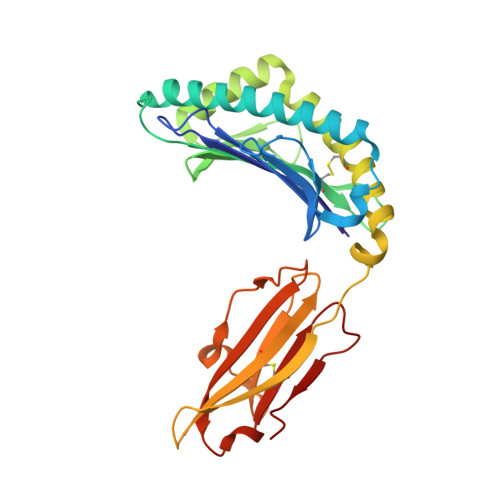
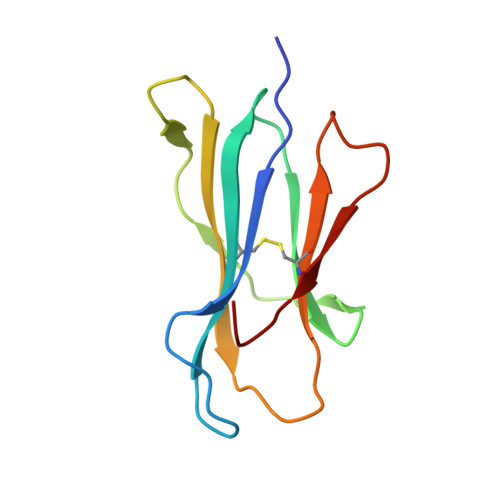
Immune self-reactivity triggered by drug-modified HLA-peptide repertoire
Illing, P.T., Vivian, J.P., Dudek, N.L., Kostenko, L., Chen, Z., Bharadwaj, M., Miles, J.J., Kjer-Nielsen, L., Gras, S., Williamson, N.A., Burrows, S.R., Purcell, A.W., Rossjohn, J., McCluskey, J.(2012) Nature 486: 554-558
- PubMed: 22722860 Search on PubMed
- DOI: https://doi.org/10.1038/nature11147
- Primary Citation Related Structures:
3VRI, 3VRJ - PubMed Abstract:
Human leukocyte antigens (HLAs) are highly polymorphic proteins that initiate immunity by presenting pathogen-derived peptides to T cells. HLA polymorphisms mostly map to the antigen-binding cleft, thereby diversifying the repertoire of self-derived and pathogen-derived peptide antigens selected by different HLA allotypes. A growing number of immunologically based drug reactions, including abacavir hypersensitivity syndrome (AHS) and carbamazepine-induced Stevens-Johnson syndrome (SJS), are associated with specific HLA alleles. However, little is known about the underlying mechanisms of these associations, including AHS, a prototypical HLA-associated drug reaction occurring exclusively in individuals with the common histocompatibility allele HLA-B*57:01, and with a relative risk of more than 1,000 (refs 6, 7). We show that unmodified abacavir binds non-covalently to HLA-B*57:01, lying across the bottom of the antigen-binding cleft and reaching into the F-pocket, where a carboxy-terminal tryptophan typically anchors peptides bound to HLA-B*57:01. Abacavir binds with exquisite specificity to HLA-B*57:01, changing the shape and chemistry of the antigen-binding cleft, thereby altering the repertoire of endogenous peptides that can bind HLA-B*57:01. In this way, abacavir guides the selection of new endogenous peptides, inducing a marked alteration in 'immunological self'. The resultant peptide-centric 'altered self' activates abacavir-specific T-cells, thereby driving polyclonal CD8 T-cell activation and a systemic reaction manifesting as AHS. We also show that carbamazepine, a widely used anti-epileptic drug associated with hypersensitivity reactions in HLA-B*15:02 individuals, binds to this allotype, producing alterations in the repertoire of presented self peptides. Our findings simultaneously highlight the importance of HLA polymorphism in the evolution of pharmacogenomics and provide a general mechanism for some of the growing number of HLA-linked hypersensitivities that involve small-molecule drugs.
- Department of Microbiology & Immunology, University of Melbourne, Parkville, Victoria 3010, Australia.
Organizational Affiliation: