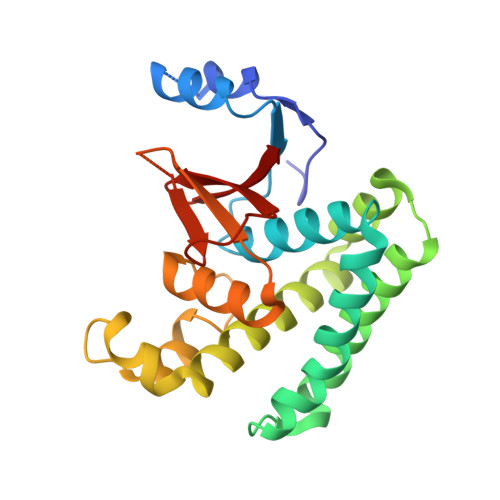
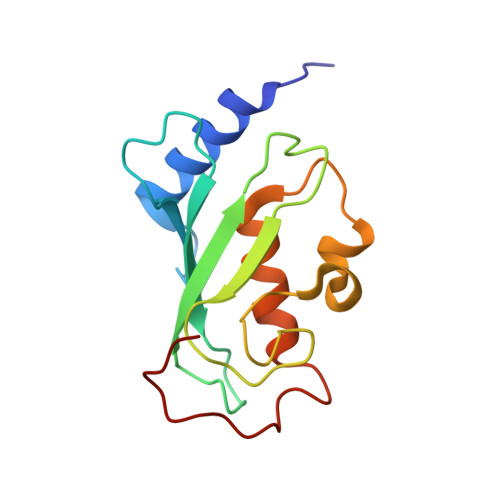
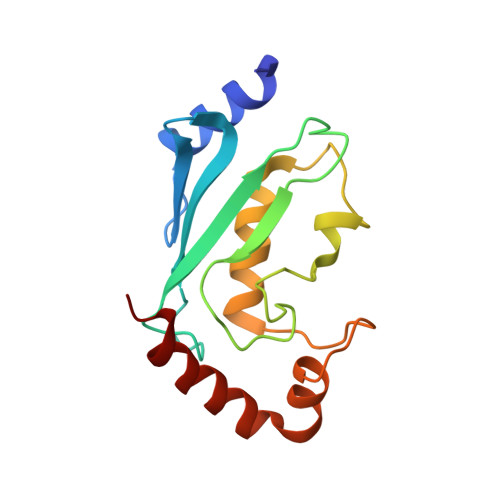
Molecular basis of Lys-63-linked polyubiquitination inhibition by the interaction between human deubiquitinating enzyme OTUB1 and ubiquitin-conjugating enzyme UBC13.
Sato, Y., Yamagata, A., Goto-Ito, S., Kubota, K., Miyamoto, R., Nakada, S., Fukai, S.(2012) J Biological Chem 287: 25860-25868
- PubMed: 22679021 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M112.364752
- Primary Citation Related Structures:
3VON - PubMed Abstract:
UBC13 is the only known E2 ubiquitin (Ub)-conjugating enzyme that produces Lys-63-linked Ub chain with its cofactor E2 variant UEV1a or MMS2. Lys-63-linked ubiquitination is crucial for recruitment of DNA repair and damage response molecules to sites of DNA double-strand breaks (DSBs). A deubiquitinating enzyme OTUB1 suppresses Lys-63-linked ubiquitination of chromatin surrounding DSBs by binding UBC13 to inhibit its E2 activity independently of the isopeptidase activity. OTUB1 strongly suppresses UBC13-dependent Lys-63-linked tri-Ub production, whereas it allows di-Ub production in vitro. The mechanism of this non-canonical OTUB1-mediated inhibition of ubiquitination remains to be elucidated. Furthermore, the atomic level information of the interaction between human OTUB1 and UBC13 has not been reported. Here, we determined the crystal structure of human OTUB1 in complex with human UBC13 and MMS2 at 3.15 Å resolution. The presented atomic-level interactions were confirmed by surface-plasmon resonance spectroscopy with structure-based mutagenesis. The designed OTUB1 mutants cannot inhibit Lys-63-linked Ub chain formation in vitro and histone ubiquitination and 53BP1 assembly around DSB sites in vivo. Finally, we propose a model for how capping of di-Ub by the OTUB1-UBC13-MMS2/UEV1a complex efficiently inhibits Lys-63-linked tri-Ub formation.
- Structural Biology Laboratory, Life Science Division, Synchrotron Radiation Research Organization and Institute of Molecular and Cellular Biosciences, University of Tokyo, Tokyo 113-0032, Japan.
Organizational Affiliation: