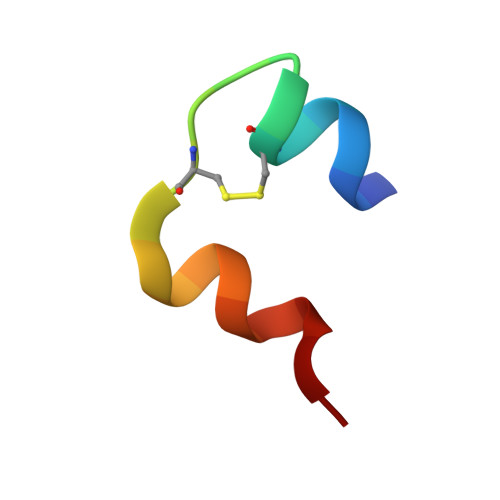
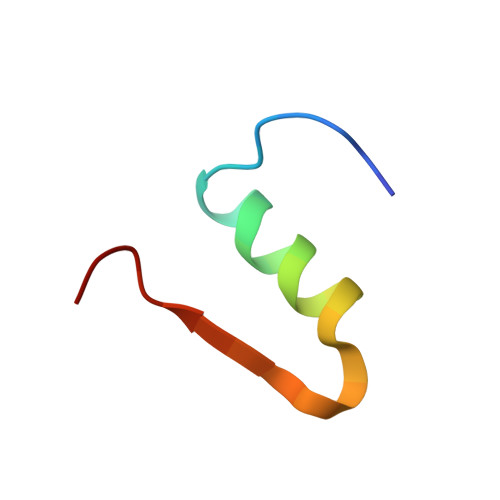
Forestalling insulin fibrillation by insertion of a chiral clamp mechanism-based application of protein engineering to global health
Wan, Z.L., Hua, Q.X., Wickramasinghe, N.P., Huang, K., Petkova, A.T., Hu, S.Q., Phillips, N.B., Yeh, I.J., Whittake, J., Ismail-Beigi, F., Katsoyyannis, P.G., Tycko, R., Weiss, M.A.To be published.