Functional and structural characterization of alpha-(1-2) branching sucrase derived from DSR-E glucansucrase
Brison, Y., Pijning, T., Malbert, Y., Fabre, E., Mourey, L., Morel, S., Potocki-Veronese, G., Monsan, P., Tranier, S., Remaud-Simeon, M., Dijkstra, B.W.(2012) J Biological Chem 287: 7915-7924
- PubMed: 22262856 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M111.305078
- Primary Citation Related Structures:
3TTO, 3TTQ - PubMed Abstract:
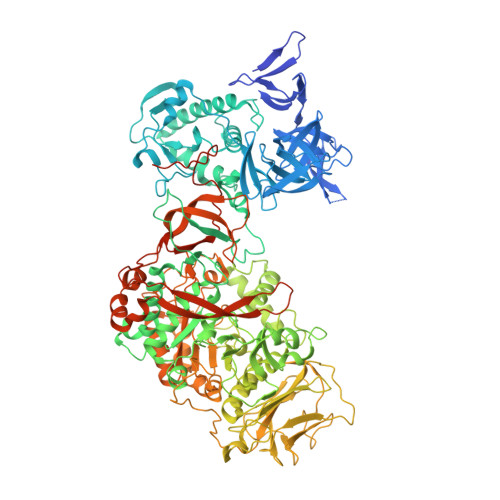
ΔN(123)-glucan-binding domain-catalytic domain 2 (ΔN(123)-GBD-CD2) is a truncated form of the bifunctional glucansucrase DSR-E from Leuconostoc mesenteroides NRRL B-1299. It was constructed by rational truncation of GBD-CD2, which harbors the second catalytic domain of DSR-E. Like GBD-CD2, this variant displays α-(1→2) branching activity when incubated with sucrose as glucosyl donor and (oligo-)dextran as acceptor, transferring glucosyl residues to the acceptor via a ping-pong bi-bi mechanism. This allows the formation of prebiotic molecules containing controlled amounts of α-(1→2) linkages. The crystal structure of the apo α-(1→2) branching sucrase ΔN(123)-GBD-CD2 was solved at 1.90 Å resolution. The protein adopts the unusual U-shape fold organized in five distinct domains, also found in GTF180-ΔN and GTF-SI glucansucrases of glycoside hydrolase family 70. Residues forming subsite -1, involved in binding the glucosyl residue of sucrose and catalysis, are strictly conserved in both GTF180-ΔN and ΔN(123)-GBD-CD2. Subsite +1 analysis revealed three residues (Ala-2249, Gly-2250, and Phe-2214) that are specific to ΔN(123)-GBD-CD2. Mutation of these residues to the corresponding residues found in GTF180-ΔN showed that Ala-2249 and Gly-2250 are not directly involved in substrate binding and regiospecificity. In contrast, mutant F2214N had lost its ability to branch dextran, although it was still active on sucrose alone. Furthermore, three loops belonging to domains A and B at the upper part of the catalytic gorge are also specific to ΔN(123)-GBD-CD2. These distinguishing features are also proposed to be involved in the correct positioning of dextran acceptor molecules allowing the formation of α-(1→2) branches.
- Université de Toulouse, INSA, UPS, INP, LISBP, F-31077 Toulouse, France.
Organizational Affiliation: