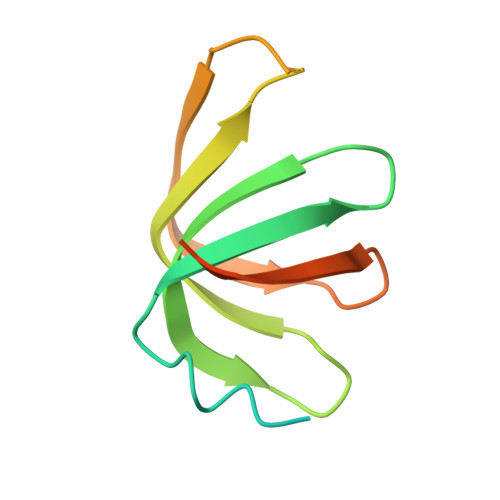
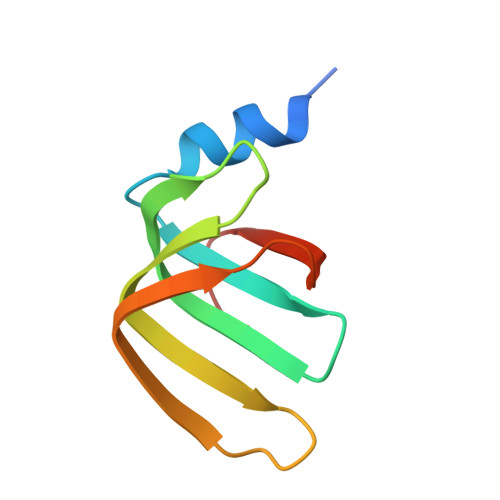
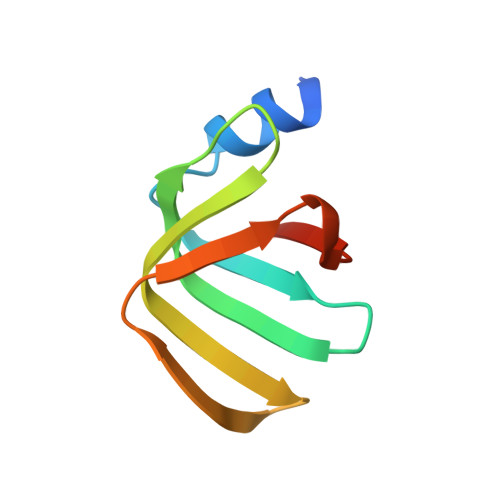
Structure of the LSm657 complex: an assembly intermediate of the LSm1-7 and LSm2-8 rings.
Mund, M., Neu, A., Ullmann, J., Neu, U., Sprangers, R.(2011) J Mol Biology 414: 165-176
- PubMed: 22001694
- DOI: https://doi.org/10.1016/j.jmb.2011.09.051
- Primary Citation Related Structures:
3SWN - PubMed Abstract:
The nuclear LSm2-8 (like Sm) complex and the cytoplasmic LSm1-7 complex play a central role in mRNA splicing and degradation, respectively. The LSm proteins are related to the spliceosomal Sm proteins that form a heteroheptameric ring around small nuclear RNA. The assembly process of the heptameric Sm complex is well established and involves several smaller Sm assembly intermediates. The assembly of the LSm complex, however, is less well studied. Here, we solved the 2.5 Å-resolution structure of the LSm assembly intermediate that contains LSm5, LSm6, and LSm7. The three monomers display the canonical Sm fold and arrange into a hexameric LSm657-657 ring. We show that the order of the LSm proteins within the ring is consistent with the order of the related SmE, SmF, and SmG proteins in the heptameric Sm ring. Nonetheless, differences in RNA binding pockets prevent the prediction of the nucleotide binding preferences of the LSm complexes. Using high-resolution NMR spectroscopy, we confirm that LSm5, LSm6, and LSm7 also assemble into a 60-kDa hexameric ring in solution. With a combination of pull-down and NMR experiments, we show that the LSm657 complex can incorporate LSm23 in order to assemble further towards native LSm rings. Interestingly, we find that the NMR spectra of the LSm57, LSm657-657, and LSm23-657 complexes differ significantly, suggesting that the angles between the LSm building blocks change depending on the ring size of the complex. In summary, our results identify LSm657 as a plastic and functional building block on the assembly route towards the LSm1-7 and LSm2-8 complexes.
- Max Planck Institute for Developmental Biology, Spemannstrasse 35, D-72076 Tuebingen, Germany.
Organizational Affiliation: