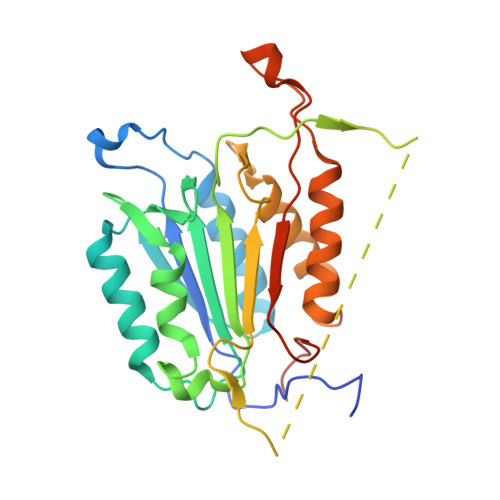
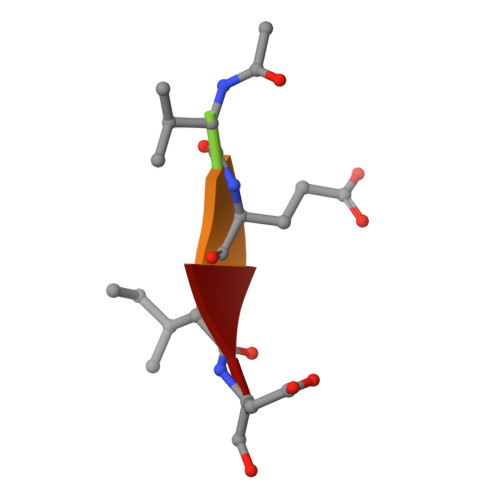
Get phases from arsenic anomalous scattering: de novo SAD phasing of two protein structures crystallized in cacodylate buffer
Liu, X., Zhang, H., Wang, X.-J., Li, L.-F., Su, X.-D.(2011) PLoS One 6: e24227-e24227
- PubMed: 21912678 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0024227
- Primary Citation Related Structures:
3S2S, 3S70 - PubMed Abstract:
The crystal structures of two proteins, a putative pyrazinamidase/nicotinamidase from the dental pathogen Streptococcus mutans (SmPncA) and the human caspase-6 (Casp6), were solved by de novo arsenic single-wavelength anomalous diffraction (As-SAD) phasing method. Arsenic (As), an uncommonly used element in SAD phasing, was covalently introduced into proteins by cacodylic acid, the buffering agent in the crystallization reservoirs. In SmPncA, the only cysteine was bound to dimethylarsinoyl, which is a pentavalent arsenic group (As (V)). This arsenic atom and a protein-bound zinc atom both generated anomalous signals. The predominant contribution, however, was from the As anomalous signals, which were sufficient to phase the SmPncA structure alone. In Casp6, four cysteines were found to bind cacodyl, a trivalent arsenic group (As (III)), in the presence of the reducing agent, dithiothreitol (DTT), and arsenic atoms were the only anomalous scatterers for SAD phasing. Analyses and discussion of these two As-SAD phasing examples and comparison of As with other traditional heavy atoms that generate anomalous signals, together with a few arsenic-based de novo phasing cases reported previously strongly suggest that As is an ideal anomalous scatterer for SAD phasing in protein crystallography.
- State Key Laboratory of Protein and Plant Gene Research and Biodynamic Optical Imaging Center (BIOPIC), School of Life Sciences, Peking University, Beijing, China.
Organizational Affiliation: