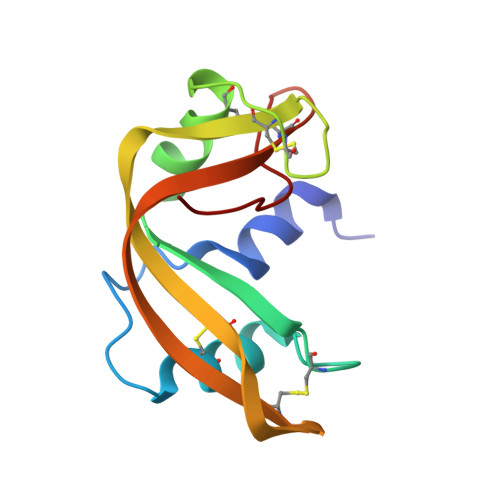
Segmented anisotropic refinement of bovine ribonuclease A by the application of the rigid-body TLS model.
Howlin, B., Moss, D.S., Harris, G.W.(1989) Acta Crystallogr A 45: 851-861
- PubMed: 2619965 Search on PubMed
- DOI: https://doi.org/10.1107/s0108767389009177
- Primary Citation Related Structures:
3RN3 - PubMed Abstract:
The anisotropic displacements of selected rigid groups in bovine ribonuclease A have been refined from X-ray diffraction data by the application of the rigid-body TLS model. The rigid groups chosen were the side chains of tyrosine, histidine and phenylalanine and the planar side chains of aspartic acid, glutamic acid, glutamine, asparagine and arginine. The method has also been applied to the co-crystallizing active-site sulfate anion. This has enabled the description of the motion of the above-mentioned side-chain atoms by anisotropic displacement ellipsoids from a 1.45 A refinement. The hydrophobic side groups in the protein core show mainly translational motion, with mean-square librations of 20 deg2 which are similar to those found in some close-packed crystals of small organic molecules. Librational displacements are much more significant in the hydrophilic side groups where their magnitudes can be correlated with solvent accessibility. Large librations of some solvent exposed side chains correspond with the breakdown of a simple TLS model and the existence of multiple orientations of the side groups. The TLS model has also been applied to the whole protein molecule and shows that the average motion is approximately isotropic with little librational character.
- Department of Crystallography, Birkbeck College, London, England.
Organizational Affiliation: