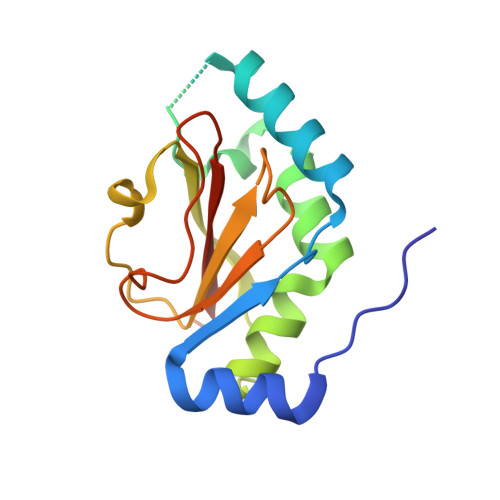
Ab initio structural modeling of and experimental validation for Chlamydia trachomatis protein CT296 reveal structural similarity to Fe(II) 2-oxoglutarate-dependent enzymes.
Kemege, K.E., Hickey, J.M., Lovell, S., Battaile, K.P., Zhang, Y., Hefty, P.S.(2011) J Bacteriol 193: 6517-6528
- PubMed: 21965559 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/JB.05488-11
- Primary Citation Related Structures:
3QH6, 3QH7 - PubMed Abstract:
Chlamydia trachomatis is a medically important pathogen that encodes a relatively high percentage of proteins with unknown function. The three-dimensional structure of a protein can be very informative regarding the protein's functional characteristics; however, determining protein structures experimentally can be very challenging. Computational methods that model protein structures with sufficient accuracy to facilitate functional studies have had notable successes. To evaluate the accuracy and potential impact of computational protein structure modeling of hypothetical proteins encoded by Chlamydia, a successful computational method termed I-TASSER was utilized to model the three-dimensional structure of a hypothetical protein encoded by open reading frame (ORF) CT296. CT296 has been reported to exhibit functional properties of a divalent cation transcription repressor (DcrA), with similarity to the Escherichia coli iron-responsive transcriptional repressor, Fur. Unexpectedly, the I-TASSER model of CT296 exhibited no structural similarity to any DNA-interacting proteins or motifs. To validate the I-TASSER-generated model, the structure of CT296 was solved experimentally using X-ray crystallography. Impressively, the ab initio I-TASSER-generated model closely matched (2.72-Å C(α) root mean square deviation [RMSD]) the high-resolution (1.8-Å) crystal structure of CT296. Modeled and experimentally determined structures of CT296 share structural characteristics of non-heme Fe(II) 2-oxoglutarate-dependent enzymes, although key enzymatic residues are not conserved, suggesting a unique biochemical process is likely associated with CT296 function. Additionally, functional analyses did not support prior reports that CT296 has properties shared with divalent cation repressors such as Fur.
- Department of Molecular Biosciences, University of Kansas, 1200 Sunnyside Avenue, Lawrence, KS 66045, USA.
Organizational Affiliation: