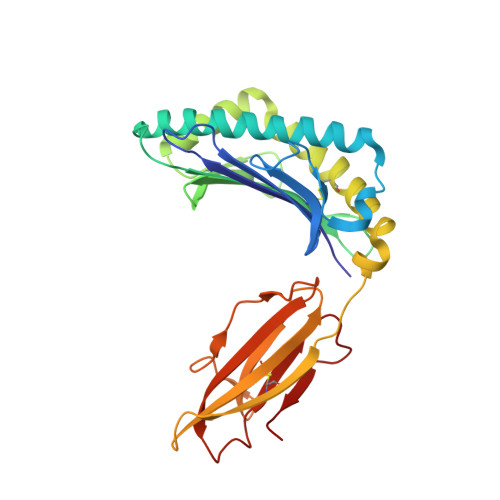
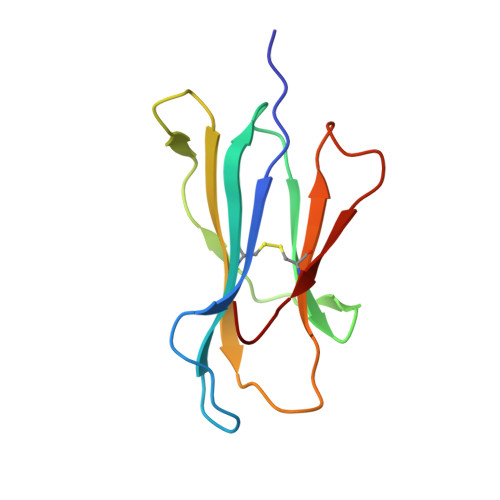
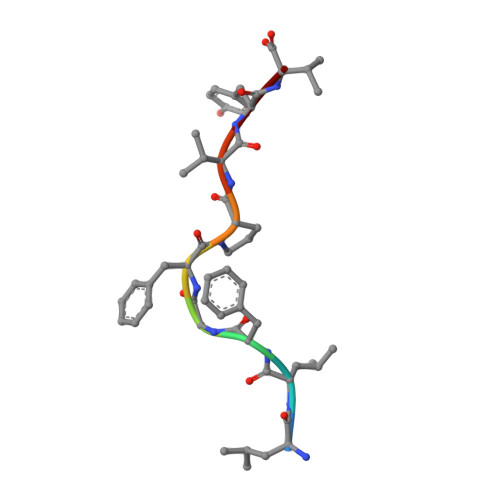
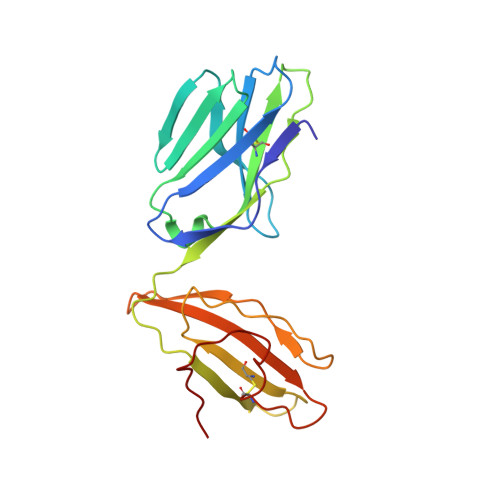
Disparate degrees of hypervariable loop flexibility control T-cell receptor cross-reactivity, specificity, and binding mechanism.
Scott, D.R., Borbulevych, O.Y., Piepenbrink, K.H., Corcelli, S.A., Baker, B.M.(2011) J Mol Biology 414: 385-400
- PubMed: 22019736 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2011.10.006
- Primary Citation Related Structures:
3QFJ, 3QH3 - PubMed Abstract:
αβ T-cell receptors (TCRs) recognize multiple antigenic peptides bound and presented by major histocompatibility complex molecules. TCR cross-reactivity has been attributed in part to the flexibility of TCR complementarity-determining region (CDR) loops, yet there have been limited direct studies of loop dynamics to determine the extent of its role. Here we studied the flexibility of the binding loops of the αβ TCR A6 using crystallographic, spectroscopic, and computational methods. A significant role for flexibility in binding and cross-reactivity was indicated only for the CDR3α and CDR3β hypervariable loops. Examination of the energy landscapes of these two loops indicated that CDR3β possesses a broad, smooth energy landscape, leading to rapid sampling in the free TCR of a range of conformations compatible with different ligands. The landscape for CDR3α is more rugged, resulting in more limited conformational sampling that leads to specificity for a reduced set of peptides as well as the major histocompatibility complex protein. In addition to informing on the mechanisms of cross-reactivity and specificity, the energy landscapes of the two loops indicate a complex mechanism for TCR binding, incorporating elements of both conformational selection and induced fit in a manner that blends features of popular models for TCR recognition.
- Department of Chemistry and Biochemistry, University of Notre Dame, Notre Dame, IN 46556, USA.
Organizational Affiliation: