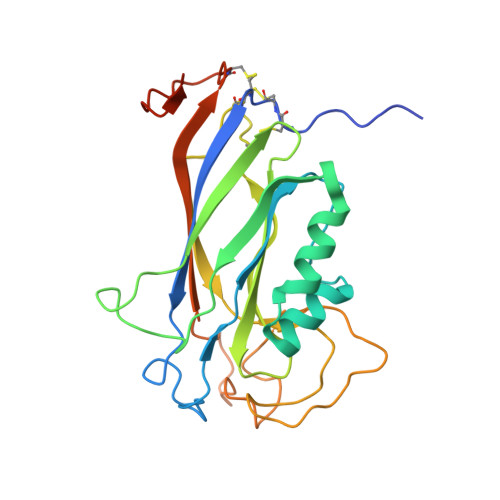
The structure of the Ca2+-binding, glycosylated F-spondin domain of F-spondin - A C2-domain variant in an extracellular matrix protein.
Tan, K., Lawler, J.(2011) BMC Struct Biol 11: 22-22
- PubMed: 21569239 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1186/1472-6807-11-22
- Primary Citation Related Structures:
3Q13 - PubMed Abstract:
F-spondin is a multi-domain extracellular matrix (ECM) protein and a contact-repellent molecule that directs axon outgrowth and cell migration during development. The reelin_N domain and the F-spondin domain (FS domain) comprise a proteolytic fragment that interacts with the cell membrane and guides the projection of commissural axons to floor plate. The FS domain is found in F-spondins, mindins, M-spondin and amphiF-spondin. We present the crystal structure of human F-spondin FS domain at 1.95Å resolution. The structure reveals a Ca2+-binding C2 domain variant with an 8-stranded antiparallel β-sandwich fold. Though the primary sequences of the FS domains of F-spondin and mindin are less than 36% identical, their overall structures are very similar. The unique feature of F-spondin FS domain is the presence of three disulfide bonds associated with the N- and C-termini of the domain and a highly conserved N-linked glycosylation site. The integrin-binding motif found in mindin is not conserved in the F-spondin FS domain. The structure of the F-spondin FS domain completes the structural studies of the multiple-domain ECM molecule. The homology of its core structure to a common Ca2+- and lipid-binding C2 domain suggests that the F-spondin FS domain may be responsible for part of the membrane targeting of F-spondin in its regulation of axon development. The structural properties of the FS domain revealed in this study pave the way for further exploration into the functions of F-spondin.
- Midwest Center for Structural Genomics and Structural Biology Center, Biosciences Division, Argonne National Laboratory, Argonne, IL 60439, USA. ktan@anl.gov
Organizational Affiliation: