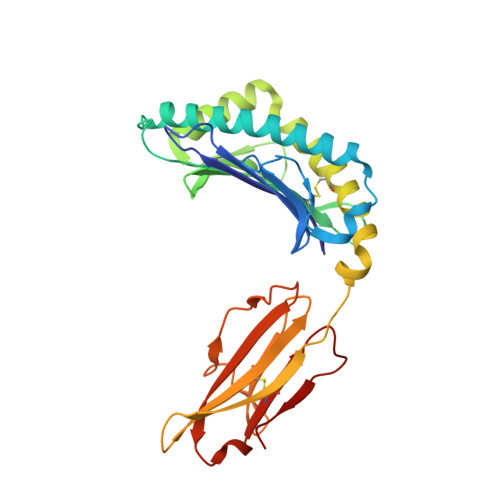
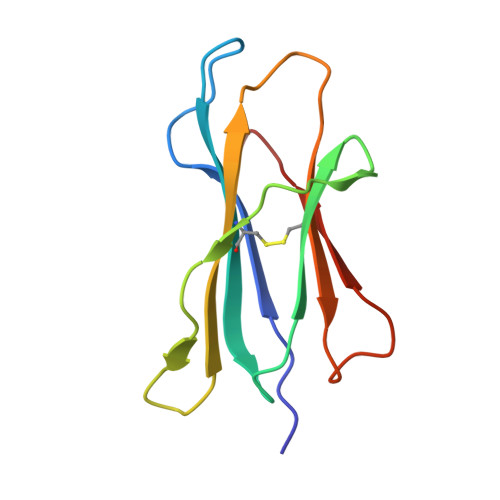
Two distinct conformations of a rinderpest virus epitope presented by bovine major histocompatibility complex class I N*01801: a host strategy to present featured peptides
Li, X., Liu, J., Qi, J., Gao, F., Li, Q., Li, X., Zhang, N., Xia, C., Gao, G.F.(2011) J Virol 85: 6038-6048
- PubMed: 21450819 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/JVI.00030-11
- Primary Citation Related Structures:
3PWU, 3PWV - PubMed Abstract:
The presentation of viral peptide epitopes to host cytotoxic T lymphocytes (CTLs) is crucial for adaptive cellular immunity to clear the virus infection, especially for some chronic viral infections. Indeed, hosts have developed effective strategies to achieve this goal. The ideal scenario would be that the peptide epitopes stimulate a broad spectrum of CTL responses with diversified T-cell receptor (TCR) usage (the TCR repertoire). It is believed that a diversified TCR repertoire requires a "featured" peptide to be presented by the host major histocompatibility complex (MHC). A featured peptide can be processed and presented in a number of ways. Here, using the X-ray diffraction method, the crystal structures of an antigenic peptide derived from rinderpest virus presented by bovine MHC class I N*01801 (BoLA-A11) have been solved, and two distinct conformations of the presented peptide are clearly displayed. A detailed analysis of the structure and comparative sequences revealed that the polymorphic amino acid isoleucine 73 (Ile73) is extremely flexible, allowing the MHC groove to adopt different conformations to accommodate the rinderpest virus peptide. This makes the peptide more featured by exposing different amino acids for T-cell recognition. The crystal structures also demonstrated that the N*01801 molecule has an unusually large A pocket, resulting in the special conformation of the P1 residue at the N terminus of the peptide. We propose that this strategy of host peptide presentation might be beneficial for creating a diversified TCR repertoire, which is important for a more-effective CTL response.
- College of Veterinary Medicine, China Agricultural University, Beijing 100094, China.
Organizational Affiliation: