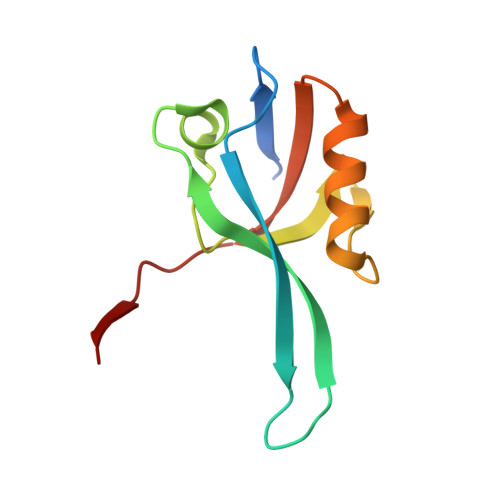
PDZ domain from Human microtubule-associated serine/threonine-protein kinase 1
Ugochukwu, E., Wang, J., Krojer, T., Muniz, J.R.C., Sethi, R., Pike, A.C.W., Roos, A., Salah, E., Cocking, R., Savitsky, P., Doyle, D.A., von Delft, F., Bountra, C., Arrowsmith, C.H., Weigelt, J., Edwards, A., Knapp, S., Elkins, J.M., Structural Genomics Consortium (SGC)To be published.