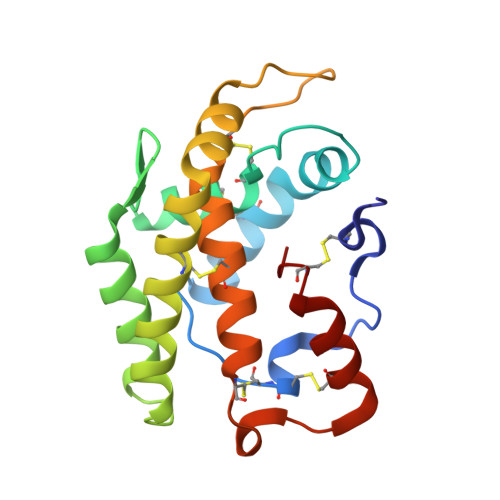
Crystal structure of a novel type of odorant-binding protein from Anopheles gambiae, belonging to the C-plus class.
Lagarde, A., Spinelli, S., Qiao, H., Tegoni, M., Pelosi, P., Cambillau, C.(2011) Biochem J 437: 423-430
- PubMed: 21561433
- DOI: https://doi.org/10.1042/BJ20110522
- Primary Citation Related Structures:
3PM2 - PubMed Abstract:
Agam (Anopheles gambiae) relies on its olfactory system to target human prey, leading eventually to the injection of Plasmodium falciparum, the malaria vector. OBPs (odorant-binding proteins) are the first line of proteins involved in odorant recognition. They interact with olfactory receptors and thus constitute an interesting target for insect control. In the present study, we undertook a large-scale analysis of proteins belonging to the olfactory system of Agam with the aim of preventing insect bites by designing strong olfactory repellents. We determined the three-dimensional structures of several Agam OBPs, either alone or in complex with model compounds. In the present paper, we report the first three-dimensional structure of a member of the C-plus class of OBPs, AgamOBP47, which has a longer sequence than classical OBPs and contains six disulfide bridges. AgamOBP47 possesses a core of six α-helices and three disulfide bridges, similar to the classical OBP fold. Two extra loops and the N- and C-terminal extra segments contain two additional α-helices and are held in conformation by three disulfide bridges. They are located either side of the classical OBP core domain. The binding site of OBP47 is located between the core and the additional domains. Two crevices are observed on opposite sides of OBP47, which are joined together by a shallow channel of sufficient size to accommodate a model of the best-tested ligand. The binding sites of C-plus class OBPs therefore exhibit different characteristics, as compared with classical OBPs, which should lead to markedly diverse functional implications.
- Architecture et Fonction des Macromolécules Biologiques, UMR 6098 CNRS and University of Marseille, 163 Av. de Luminy Case 932, 13288 Marseille Cedex 09, France.
Organizational Affiliation: