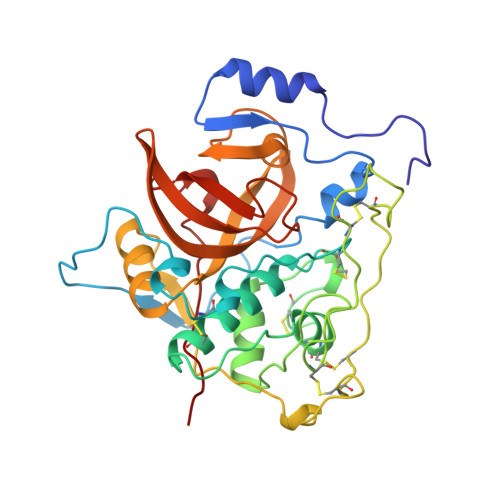
Crystal structure of the wild-type human procathepsin B at 2.5 A resolution reveals the native active site of a papain-like cysteine protease zymogen.
Podobnik, M., Kuhelj, R., Turk, V., Turk, D.(1997) J Mol Biology 271: 774-788
- PubMed: 9299326 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1997.1218
- Primary Citation Related Structures:
3PBH - PubMed Abstract:
The structure of the wild-type human procathepsin B has been refined to a crystallographic R-value of 0.18 and R-free of 0.23 exploiting the data obtained from new crystals that diffract beyond 2.5 A resolution. The structure confirms two previously presented, lower-resolution structures. The structure of the propeptide chain folds on the surface of the enzyme domains and blocks access of substrate to the already formed active site. Abundant solvent molecules fill the cavities between the propeptide and the enzyme part of the molecule. The propeptide structure is compared with a substrate model in the S2, S1, S1' and S2' binding sites. In this crystal form the cathepsin B occluding loop residues adopt yet another conformation. The structures show that the occluding loop region between the residues Cys108 and Cys119 behaves quite independently from the rest of the structure and easily adapts to changes in environment. The variety of the observed conformations of the occluding loop is in agreement with other data showing that the loop is responsible for limiting cathepsin B activity to that of a carboxydipeptidase. The region before Cys108 is essentially the same as in the mature structure, whereas the region from Cys119 to Thr125 is raised compared to the mature form by the propeptide squeezed between it and the enzyme domains, surface. The structure strongly suggests that processing of procathepsin B during its autoactivation is not unimolecular.
- Department of Biochemistry and Molecular Biology, Jozef Stefan Institute, Jamova 39, Ljubljana, 1111, Slovenia.
Organizational Affiliation: