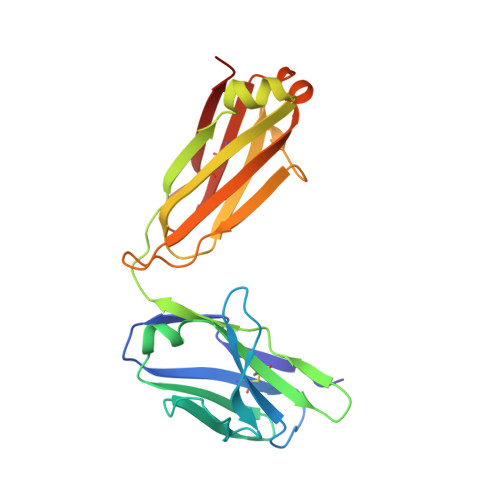
A nonself sugar mimic of the HIV glycan shield shows enhanced antigenicity.
Doores, K.J., Fulton, Z., Hong, V., Patel, M.K., Scanlan, C.N., Wormald, M.R., Finn, M.G., Burton, D.R., Wilson, I.A., Davis, B.G.(2010) Proc Natl Acad Sci U S A 107: 17107-17112
- PubMed: 20852065 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1002717107
- Primary Citation Related Structures:
3OAY, 3OAZ, 3OB0 - PubMed Abstract:
Antibody 2G12 uniquely neutralizes a broad range of HIV-1 isolates by binding the high-mannose glycans on the HIV-1 surface glycoprotein, gp120. Antigens that resemble these natural epitopes of 2G12 would be highly desirable components for an HIV-1 vaccine. However, host-produced (self)-carbohydrate motifs have been unsuccessful so far at eliciting 2G12-like antibodies that cross-react with gp120. Based on the surprising observation that 2G12 binds nonproteinaceous monosaccharide D-fructose with higher affinity than D-mannose, we show here that a designed set of nonself, synthetic monosaccharides are potent antigens. When introduced to the terminus of the D1 arm of protein glycans recognized by 2G12, their antigenicity is significantly enhanced. Logical variation of these unnatural sugars pinpointed key modifications, and the molecular basis of this increased antigenicity was elucidated using high-resolution crystallographic analyses. Virus-like particle protein conjugates containing such nonself glycans are bound more tightly by 2G12. As immunogens they elicit higher titers of antibodies than those immunogenic conjugates containing the self D1 glycan motif. These antibodies generated from nonself immunogens also cross-react with this self motif, which is found in the glycan shield, when it is presented in a range of different conjugates and glycans. However, these antibodies did not bind this glycan motif when present on gp120.
- Department of Chemistry, University of Oxford, Chemistry Research Laboratory, Mansfield Road, Oxford, United Kingdom.
Organizational Affiliation: