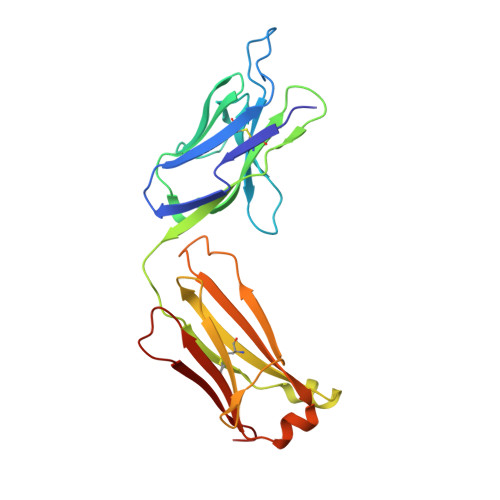
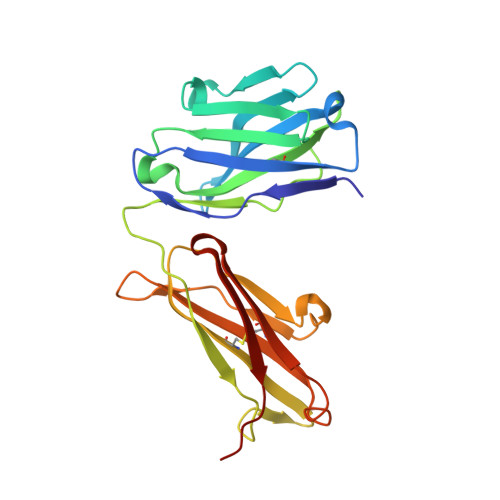
Structural basis of clade-specific HIV-1 neutralization by humanized anti-V3 monoclonal antibody KD-247.
Kirby, K.A., Ong, Y.T., Hachiya, A., Laughlin, T.G., Chiang, L.A., Pan, Y., Moran, J.L., Marchand, B., Singh, K., Gallazzi, F., Quinn, T.P., Yoshimura, K., Murakami, T., Matsushita, S., Sarafianos, S.G.(2015) FASEB J 29: 70-80
- PubMed: 25351987 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1096/fj.14-252262
- Primary Citation Related Structures:
3NTC - PubMed Abstract:
Humanized monoclonal antibody KD-247 targets the Gly(312)-Pro(313)-Gly(314)-Arg(315) arch of the third hypervariable (V3) loop of the HIV-1 surface glycoprotein. It potently neutralizes many HIV-1 clade B isolates, but not of other clades. To understand the molecular basis of this specificity, we solved a high-resolution (1.55 Å) crystal structure of the KD-247 antigen binding fragment and examined the potential interactions with various V3 loop targets. Unlike most antibodies, KD-247 appears to interact with its target primarily through light chain residues. Several of these interactions involve Arg(315) of the V3 loop. To evaluate the role of light chain residues in the recognition of the V3 loop, we generated 20 variants of KD-247 single-chain variable fragments with mutations in the antigen-binding site. Purified proteins were assessed for V3 loop binding using AlphaScreen technology and for HIV-1 neutralization. Our data revealed that recognition of the clade-specificity defining residue Arg(315) of the V3 loop is based on a network of interactions that involve Tyr(L32), Tyr(L92), and Asn(L27d) that directly interact with Arg(315), thus elucidating the molecular interactions of KD-247 with its V3 loop target.
- Christopher S. Bond Life Sciences Center, Department of Molecular Microbiology and Immunology, School of Medicine.
Organizational Affiliation: