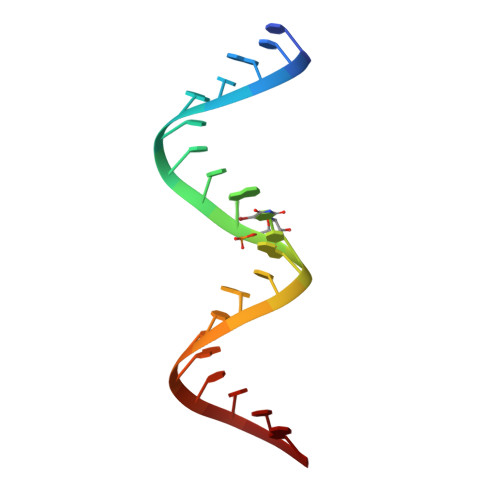
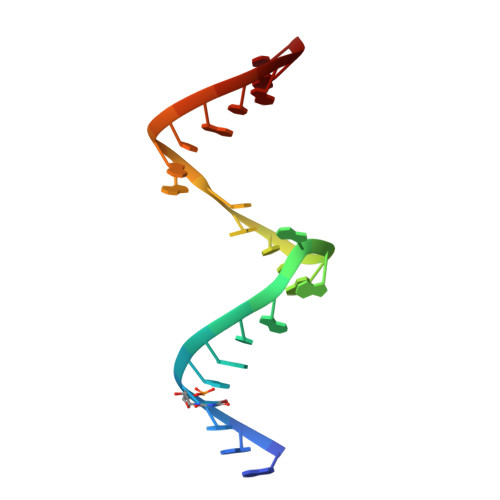
A model for the study of ligand binding to the ribosomal RNA helix h44.
Dibrov, S.M., Parsons, J., Hermann, T.(2010) Nucleic Acids Res 38: 4458-4465
- PubMed: 20215440 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkq159
- Primary Citation Related Structures:
3LOA - PubMed Abstract:
Oligonucleotide models of ribosomal RNA domains are powerful tools to study the binding and molecular recognition of antibiotics that interfere with bacterial translation. Techniques such as selective chemical modification, fluorescence labeling and mutations are cumbersome for the whole ribosome but readily applicable to model RNAs, which are readily crystallized and often give rise to higher resolution crystal structures suitable for detailed analysis of ligand-RNA interactions. Here, we have investigated the HX RNA construct which contains two adjacent ligand binding regions of helix h44 in 16S ribosomal RNA. High-resolution crystal structure analysis confirmed that the HX RNA is a faithful structural model of the ribosomal target. Solution studies showed that HX RNA carrying a fluorescent 2-aminopurine modification provides a model system that can be used to monitor ligand binding to both the ribosomal decoding site and, through an indirect effect, the hygromycin B interaction region.
- Department of Chemistry and Biochemistry, University of California, San Diego, 9500 Gilman Drive, La Jolla, CA 92093, USA.
Organizational Affiliation: