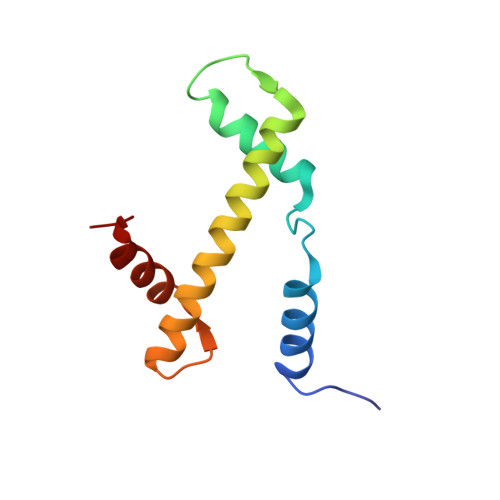
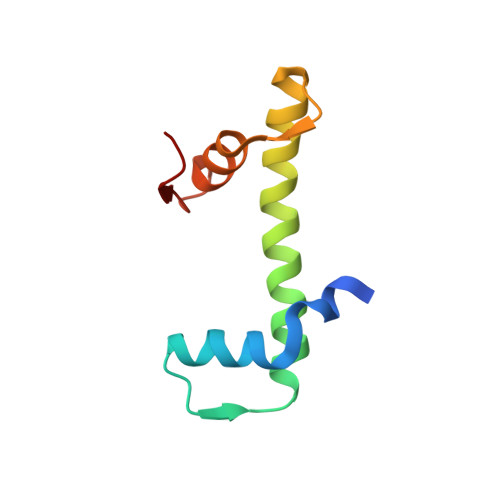
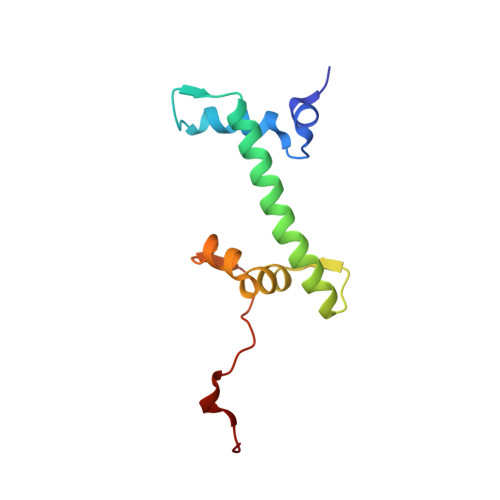
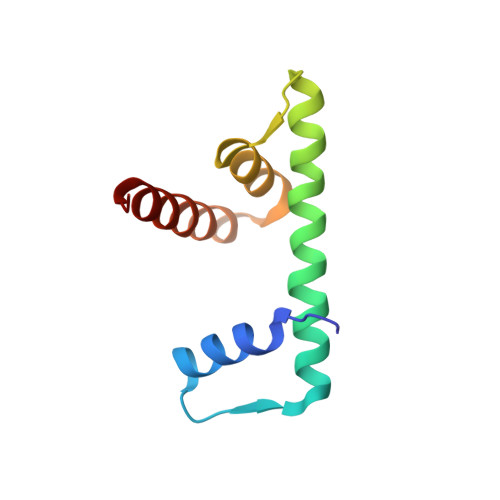
Structural characterization of H3K56Q nucleosomes and nucleosomal arrays.
Watanabe, S., Resch, M., Lilyestrom, W., Clark, N., Hansen, J.C., Peterson, C., Luger, K.(null) Biochim Biophys Acta 1799: 480-486
- PubMed: 20100606 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.bbagrm.2010.01.009
- Primary Citation Related Structures:
3KWQ, 3KXB - PubMed Abstract:
The post-translational modification of histones is a key mechanism for the modulation of DNA accessibility. Acetylated lysine 56 in histone H3 is associated with nucleosome assembly during replication and DNA repair, and is thus likely to predominate in regions of chromatin containing nucleosome-free regions. Here we show by X-ray crystallography that mutation of H3 lysine 56 to glutamine (to mimic acetylation) or glutamate (to cause a charge reversal) has no detectable effects on the structure of the nucleosome. At the level of higher order chromatin structure, the K to Q substitution has no effect on the folding of model nucleosomal arrays in cis, regardless of the degree of nucleosome density. In contrast, defects in array-array interactions in trans ('oligomerization') are selectively observed for mutant H3 lysine 56 arrays that contain nucleosome-free regions. Our data suggests that H3K56 acetylation is one of the molecular mechanisms employed to keep chromatin with nucleosome-free regions accessible to the DNA replication and repair machinery.
- Program in Molecular Medicine, University of Massachusetts Medical School, 373 Plantation St., Worcester, MA 01605, USA.
Organizational Affiliation: