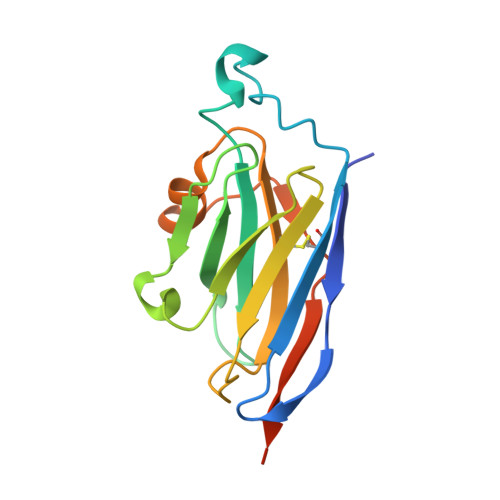
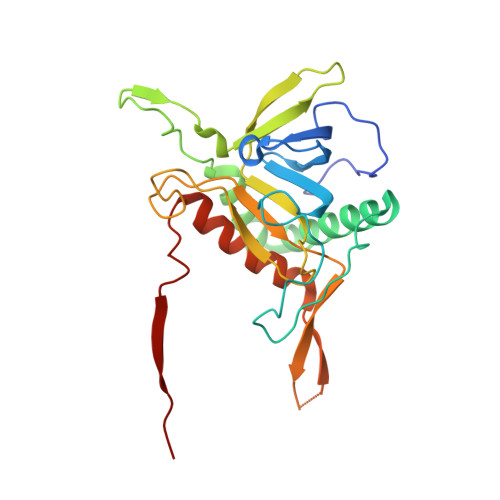
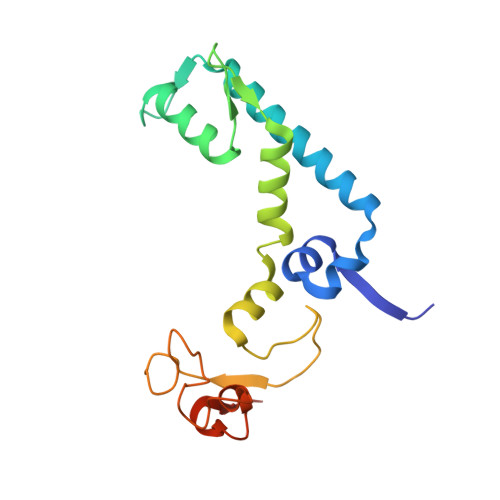
A Single-Domain Llama Antibody Potently Inhibits the Enzymatic Activity of Botulinum Neurotoxin by Binding to the Non-Catalytic alpha-Exosite Binding Region.
Dong, J., Thompson, A.A., Fan, Y., Lou, J., Conrad, F., Ho, M., Pires-Alves, M., Wilson, B.A., Stevens, R.C., Marks, J.D.(2010) J Mol Biology 397: 1106-1118
- PubMed: 20138889 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2010.01.070
- Primary Citation Related Structures:
3K3Q - PubMed Abstract:
Ingestion or inhalation of botulinum neurotoxin (BoNT) results in botulism, a severe and frequently fatal disease. Current treatments rely on antitoxins, which, while effective, cannot reverse symptoms once BoNT has entered the neuron. For treatments that can reverse intoxication, interest has focused on developing inhibitors of the enzymatic BoNT light chain (BoNT Lc). Such inhibitors typically mimic substrate and bind in or around the substrate cleavage pocket. To explore the full range of binding sites for serotype A light chain (BoNT/A Lc) inhibitors, we created a library of non-immune llama single-domain VHH (camelid heavy-chain variable region derived from heavy-chain-only antibody) antibodies displayed on the surface of the yeast Saccharomyces cerevisiae. Library selection on BoNT/A Lc yielded 15 yeast-displayed VHH with equilibrium dissociation constants (K(d)) from 230 to 0.03 nM measured by flow cytometry. Eight of 15 VHH inhibited the cleavage of substrate SNAP25 (synaptosome-associated protein of 25,000 Da) by BoNT/A Lc. The most potent VHH (Aa1) had a solution K(d) for BoNT/A Lc of 1.47 x 10(-)(10) M and an IC(50) (50% inhibitory concentration) of 4.7 x 10(-)(10) M and was resistant to heat denaturation and reducing conditions. To understand the mechanism by which Aa1 inhibited catalysis, we solved the X-ray crystal structure of the BoNT/A Lc-Aa1 VHH complex at 2.6 A resolution. The structure reveals that the Aa1 VHH binds in the alpha-exosite of the BoNT/A Lc, far from the active site for catalysis. The study validates the utility of non-immune llama VHH libraries as a source of enzyme inhibitors and identifies the BoNT/A Lc alpha-exosite as a target for inhibitor development.
- Department of Anesthesia, University of California, San Francisco, San Francisco, CA 94110, USA.
Organizational Affiliation: