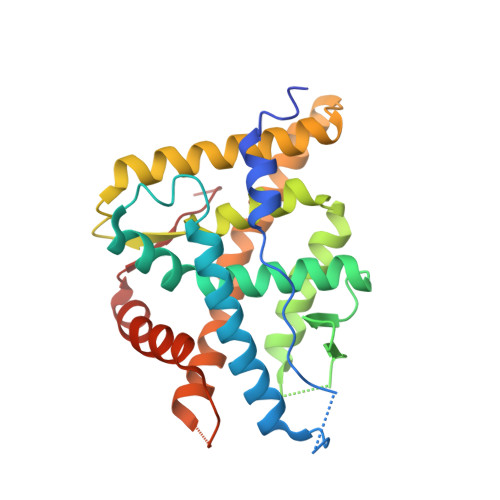
Design and x-ray crystal structures of high-potency nonsteroidal glucocorticoid agonists exploiting a novel binding site on the receptor.
Biggadike, K., Bledsoe, R.K., Coe, D.M., Cooper, T.W., House, D., Iannone, M.A., Macdonald, S.J., Madauss, K.P., McLay, I.M., Shipley, T.J., Taylor, S.J., Tran, T.B., Uings, I.J., Weller, V., Williams, S.P.(2009) Proc Natl Acad Sci U S A 106: 18114-18119
- PubMed: 19822747 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0909125106
- Primary Citation Related Structures:
3K22, 3K23 - PubMed Abstract:
Crystallography and computer modeling have been used to exploit a previously unexplored channel in the glucocorticoid receptor (GR). Highly potent, nonsteroidal indazole amides showing excellent complementarity to the channel were designed with the assistance of the computational technique AlleGrow. The accuracy of the design process was demonstrated through crystallographic structural determination of the GR ligand-binding domain-agonist complex of the D-prolinamide derivative 11. The utility of the channel was further exemplified through the design of a potent phenylindazole in which structural motifs, seen to interact with the traditional GR ligand pocket, were abandoned and replaced by interactions within the new channel. Occupation of the channel was confirmed with a second GR crystal structure of this truncated D-alaninamide derivative 13. Compound 11 displays properties compatible with development as an intranasal solution formulation, whereas oral bioavailability has been demonstrated with a related truncated exemplar 14. Data with the pyrrolidinone amide 12 demonstrate the potential for further elaboration within the "meta" channel to deliver compounds with selectivity for the desired transrepressive activity of glucocorticoids. The discovery of these interactions with this important receptor offers significant opportunities for the design of novel GR modulators.
- Medicinal Chemistry, Respiratory Center of Excellence for Drug Discovery, GlaxoSmithKline Medicines Research Center, Stevenage, Hertfordshire SG1 2NY, United Kingdom. keith.b.biggadike@gsk.com
Organizational Affiliation: