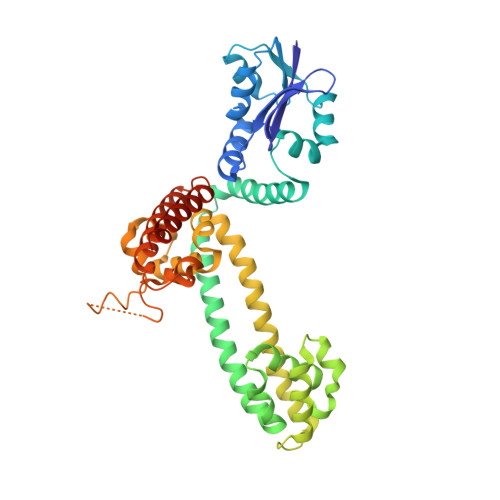
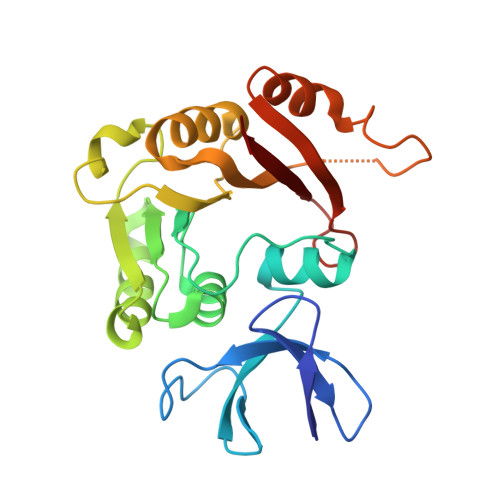
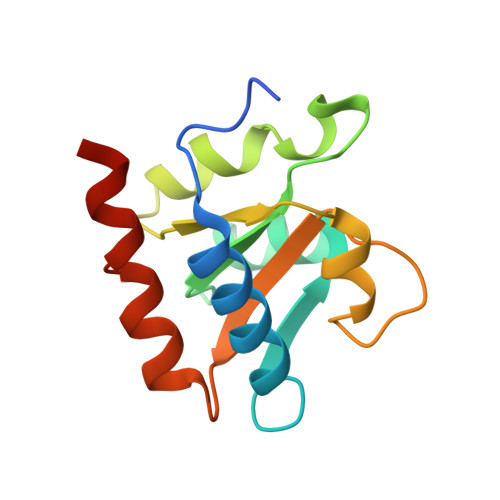
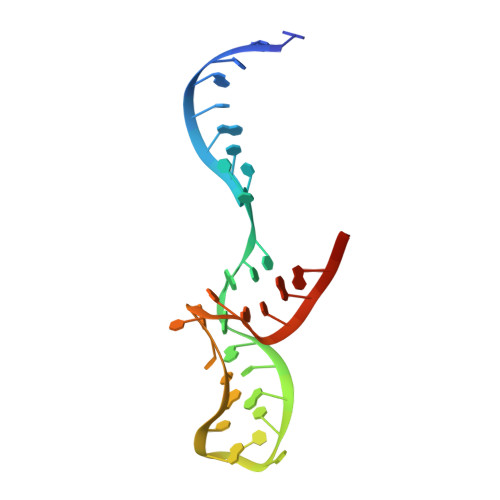
Structural organization of box C/D RNA-guided RNA methyltransferase.
Ye, K., Jia, R., Lin, J., Ju, M., Peng, J., Xu, A., Zhang, L.(2009) Proc Natl Acad Sci U S A 106: 13808-13813
- PubMed: 19666563 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0905128106
- Primary Citation Related Structures:
3ICX, 3ID5, 3ID6 - PubMed Abstract:
Box C/D guide RNAs are abundant noncoding RNAs that primarily function to direct the 2'-O-methylation of specific nucleotides by base-pairing with substrate RNAs. In archaea, a bipartite C/D RNA assembles with L7Ae, Nop5, and the methyltransferase fibrillarin into a modification enzyme with unique substrate specificity. Here, we determined the crystal structure of an archaeal C/D RNA-protein complex (RNP) composed of all 3 core proteins and an engineered half-guide RNA at 4 A resolution, as well as 2 protein substructures at higher resolution. The RNP structure reveals that the C-terminal domains of Nop5 in the dimeric complex provide symmetric anchoring sites for 2 L7Ae-associated kink-turn motifs of the C/D RNA. A prominent protrusion in Nop5 seems to be important for guide RNA organization and function and for discriminating the structurally related U4 snRNA. Multiple conformations of the N-terminal domain of Nop5 and its associated fibrillarin in different structures indicate the inherent flexibility of the catalytic module, suggesting that a swinging motion of the catalytic module is part of the enzyme mechanism. We also built a model of a native C/D RNP with substrate and fibrillarin in an active conformation. Our results provide insight into the overall organization and mechanism of action of C/D RNA-guided RNA methyltransferases.
- National Institute of Biological Sciences, Beijing 102206, China.
Organizational Affiliation: