crystal structure of human BFL-1 in complex with BAK BH3 peptide
Guan, R., Xiao, R., Zhao, L., Acton, T., White, E., Gelinas, C., Montelione, G.T.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein BFL-1 | 161 | Homo sapiens | Mutation(s): 0 Gene Names: BCL2A1, BCL2L5, BFL1, GRS, HBPA1 | 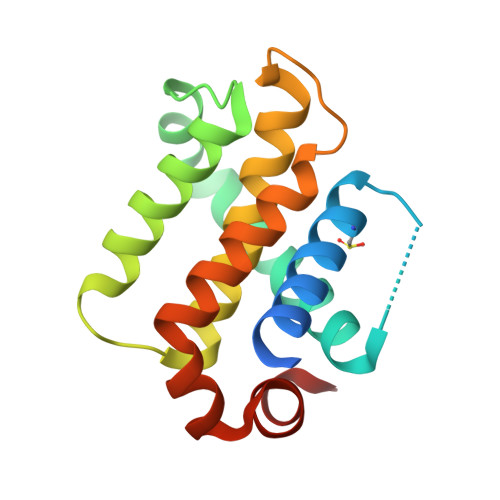 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q16548 GTEx: ENSG00000140379 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q16548 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Apoptosis regulator BAK | 16 | Homo sapiens | Mutation(s): 0 |  | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q16611 GTEx: ENSG00000030110 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q16611 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CSD Query on CSD | A | L-PEPTIDE LINKING | C3 H7 N O4 S |  | CYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 43.236 | α = 90 |
| b = 43.145 | β = 114.45 |
| c = 46.068 | γ = 90 |
| Software Name | Purpose |
|---|---|
| MAR345dtb | data collection |
| AMoRE | phasing |
| CNS | refinement |
| CrystalClear | data reduction |
| CrystalClear | data scaling |