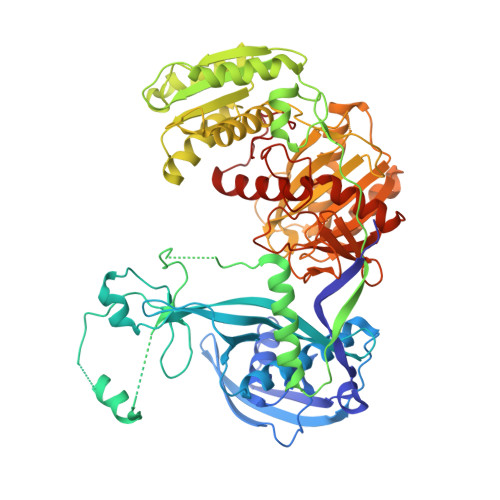
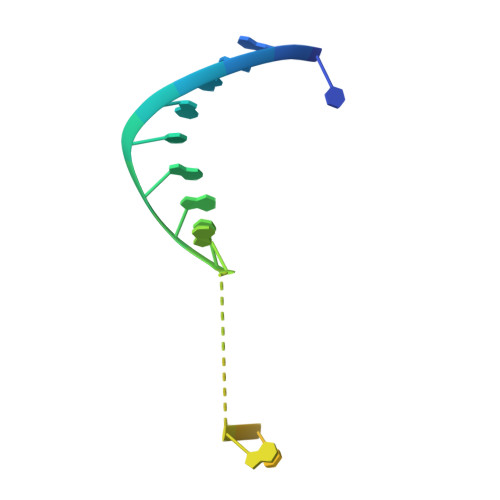
Nucleation, propagation and cleavage of target RNAs in Ago silencing complexes.
Wang, Y., Juranek, S., Li, H., Sheng, G., Wardle, G.S., Tuschl, T., Patel, D.J.(2009) Nature 461: 754-761
- PubMed: 19812667 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nature08434
- Primary Citation Related Structures:
3HJF, 3HK2, 3HM9, 3HO1, 3HVR, 3HXM - PubMed Abstract:
The slicer activity of the RNA-induced silencing complex resides within its Argonaute (Ago) component, in which the PIWI domain provides the catalytic residues governing guide-strand mediated site-specific cleavage of target RNA. Here we report on structures of ternary complexes of Thermus thermophilus Ago catalytic mutants with 5'-phosphorylated 21-nucleotide guide DNA and complementary target RNAs of 12, 15 and 19 nucleotides in length, which define the molecular basis for Mg(2+)-facilitated site-specific cleavage of the target. We observe pivot-like domain movements within the Ago scaffold on proceeding from nucleation to propagation steps of guide-target duplex formation, with duplex zippering beyond one turn of the helix requiring the release of the 3'-end of the guide from the PAZ pocket. Cleavage assays on targets of various lengths supported this model, and sugar-phosphate-backbone-modified target strands showed the importance of structural and catalytic divalent metal ions observed in the crystal structures.
- Structural Biology Program, Memorial-Sloan Kettering Cancer Center, New York, New York 10065, USA.
Organizational Affiliation: