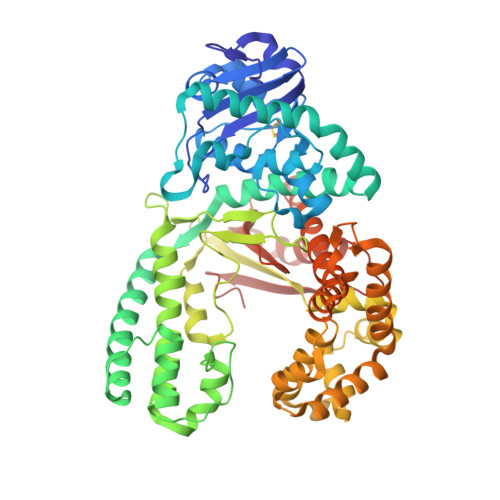
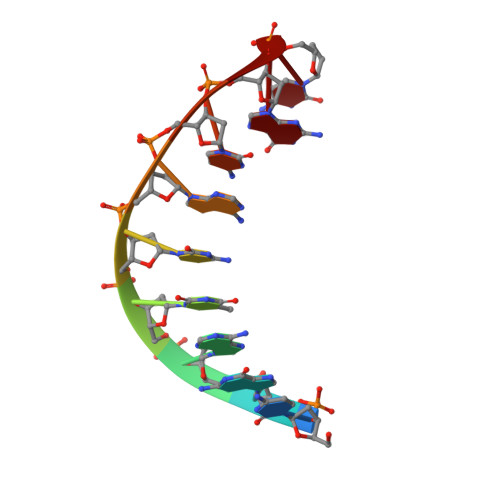
The structure of a high fidelity DNA polymerase bound to a mismatched nucleotide reveals an "ajar" intermediate conformation in the nucleotide selection mechanism.
Wu, E.Y., Beese, L.S.(2011) J Biological Chem 286: 19758-19767
- PubMed: 21454515 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M110.191130
- Primary Citation Related Structures:
3HP6, 3HPO, 3HT3 - PubMed Abstract:
To achieve accurate DNA synthesis, DNA polymerases must rapidly sample and discriminate against incorrect nucleotides. Here we report the crystal structure of a high fidelity DNA polymerase I bound to DNA primer-template caught in the act of binding a mismatched (dG:dTTP) nucleoside triphosphate. The polymerase adopts a conformation in between the previously established "open" and "closed" states. In this "ajar" conformation, the template base has moved into the insertion site but misaligns an incorrect nucleotide relative to the primer terminus. The displacement of a conserved active site tyrosine in the insertion site by the template base is accommodated by a distinctive kink in the polymerase O helix, resulting in a partially open ternary complex. We suggest that the ajar conformation allows the template to probe incoming nucleotides for complementarity before closure of the enzyme around the substrate. Based on solution fluorescence, kinetics, and crystallographic analyses of wild-type and mutant polymerases reported here, we present a three-state reaction pathway in which nucleotides either pass through this intermediate conformation to the closed conformation and catalysis or are misaligned within the intermediate, leading to destabilization of the closed conformation.
- Department of Biochemistry, Duke University Medical Center, Durham, North Carolina 27710, USA.
Organizational Affiliation: