Macrophage Inflammatory Protein-1 Is A Novel High Affinity Substrate For Human Insulin Degrading Enzyme
Ren, M., Guo, Q., Tang, W.J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Insulin-degrading enzyme | 990 | Homo sapiens | Mutation(s): 14 Gene Names: IDE EC: 3.4.24.56 | 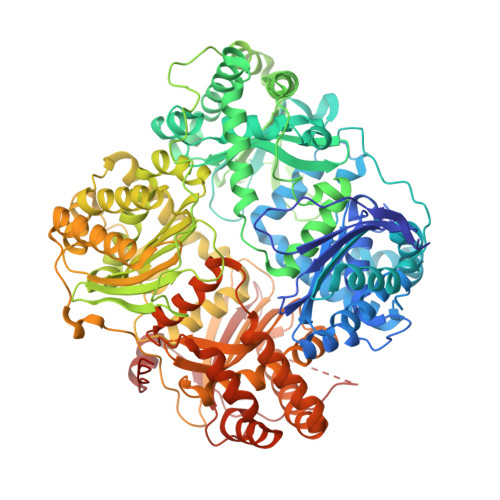 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P14735 GTEx: ENSG00000119912 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P14735 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| C-C motif chemokine 3 | 70 | Homo sapiens | Mutation(s): 0 Gene Names: CCL3, G0S19-1, MIP1A, MIP1alpha, SCYA3 | 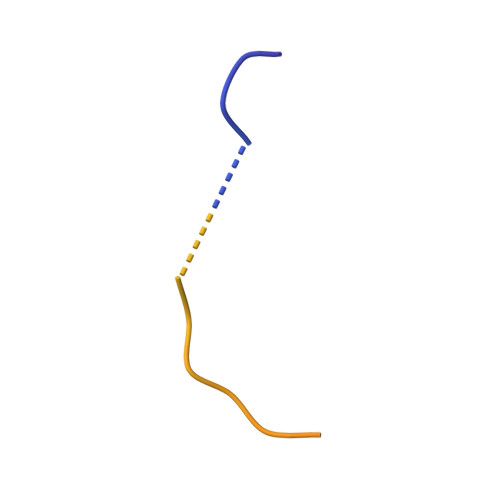 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P10147 GTEx: ENSG00000277632 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P10147 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| DIO Download:Ideal Coordinates CCD File | E [auth A] F [auth A] G [auth A] I [auth B] J [auth B] | 1,4-DIETHYLENE DIOXIDE C4 H8 O2 RYHBNJHYFVUHQT-UHFFFAOYSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | H [auth B], L [auth C] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 262.737 | α = 90 |
| b = 262.737 | β = 90 |
| c = 90.502 | γ = 120 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data collection |
| PHASES | phasing |
| REFMAC | refinement |
| HKL-2000 | data reduction |
| SCALEPACK | data scaling |