X-ray crystal structure of peptide-binding domain of Kar2 protein from Saccharomyces cerevisiae.
Osipiuk, J., Bigelow, L., Gu, M., Sahi, C., Craig, E.A., Joachimiak, A.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 78 kDa glucose-regulated protein homolog | 152 | Saccharomyces cerevisiae | Mutation(s): 0 Gene Names: GRP78, J1248, KAR2, SSD1, YJL034W EC: 3.6.4.10 | 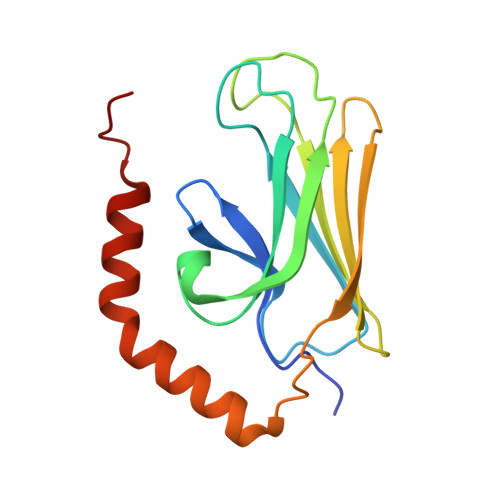 | |
UniProt | |||||
Find proteins for P16474 (Saccharomyces cerevisiae (strain ATCC 204508 / S288c)) Explore P16474 Go to UniProtKB: P16474 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P16474 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 41.846 | α = 90 |
| b = 103.113 | β = 90 |
| c = 80.792 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALEPACK | data scaling |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| SBC-Collect | data collection |
| HKL-3000 | data reduction |
| HKL-3000 | phasing |
| MOLREP | phasing |