The molecular structure of wild-type and a mutant Fis protein: relationship between mutational changes and recombinational enhancer function or DNA binding.
Yuan, H.S., Finkel, S.E., Feng, J.A., Kaczor-Grzeskowiak, M., Johnson, R.C., Dickerson, R.E.(1991) Proc Natl Acad Sci U S A 88: 9558-9562
- PubMed: 1946369 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.88.21.9558
- Primary Citation Related Structures:
3FIS, 4FIS - PubMed Abstract:
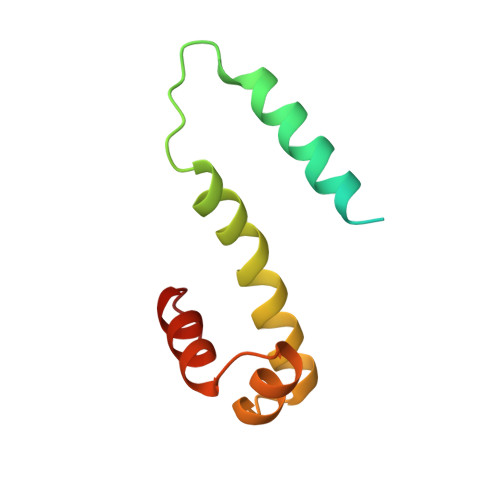
The 98-amino acid Fis protein from Escherichia coli functions in a variety of reactions, including promotion of Hin-mediated site-specific DNA inversion when bound to an enhancer sequence. It is unique among site-specific DNA-binding proteins in that it binds to a large number of different DNA sequences, for which a consensus sequence is difficult to establish. X-ray crystal structure analyses have been carried out at 2.3 A resolution for wild-type Fis and for an Arg-89----Cys mutant that does not stimulate DNA inversion. Each monomer of the Fis dimer has four alpha-helices, A-D; the first 19 residues are disordered in the crystal. The end of each C helix is hydrogen bonded to the beginning of helix B' from the opposite subunit in what effectively is one long continuous, although bent, helix. The four helices, C, B', C', and B, together define a platform through the center of the Fis molecule: helices A and A' are believed to be involved with Hin recombinase on one side, and helices D and D' interact with DNA lying on the other side of the platform. Helices C and D of each subunit comprise a helix-turn-helix (HTH) DNA-binding element. The spacing of these two HTH elements in the dimer, 25 A, is too short to allow insertion into adjacent major grooves of a straight B-DNA helix. However, bending the DNA at discrete points, to an overall radius of curvature of 62 A, allows efficient docking of a B-DNA helix with the Fis molecule. The proposed complex explains the experimentally observed patterns of methylation protection and DNase I cleavage hypersensitivity. The x-ray structure accounts for the effects of mutations in the Fis sequence. Those that affect DNA inversion but not DNA binding are located within the N-terminal disordered region and helix A. This inversion activation domain is physically separated in the Fis molecule from the HTH elements and may specify a region of contact with the Hin recombinase. In contrast, mutations that affect HTH helices C and D, or interactions of these with helix B, have the additional effect of decreasing or eliminating binding to DNA.
- Molecular Biology Institute, University of California, Los Angeles 90024.
Organizational Affiliation: