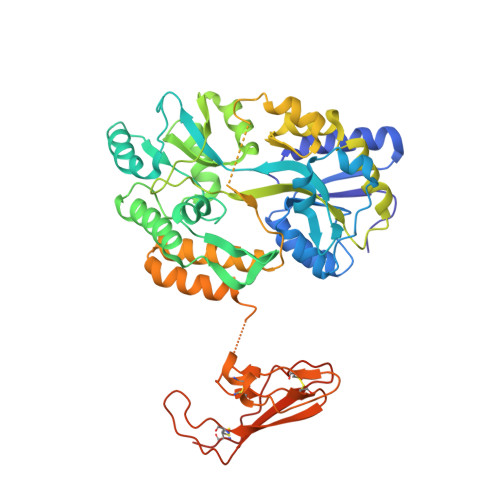
Molecular Recognition of Corticotropin-releasing Factor by Its G-protein-coupled Receptor CRFR1.
Pioszak, A.A., Parker, N.R., Suino-Powell, K., Xu, H.E.(2008) J Biological Chem 283: 32900-32912
- PubMed: 18801728 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M805749200
- Primary Citation Related Structures:
3EHS, 3EHT, 3EHU - PubMed Abstract:
The bimolecular interaction between corticotropin-releasing factor (CRF), a neuropeptide, and its type 1 receptor (CRFR1), a class B G-protein-coupled receptor (GPCR), is crucial for activation of the hypothalamic-pituitary-adrenal axis in response to stress, and has been a target of intense drug design for the treatment of anxiety, depression, and related disorders. As a class B GPCR, CRFR1 contains an N-terminal extracellular domain (ECD) that provides the primary ligand binding determinants. Here we present three crystal structures of the human CRFR1 ECD, one in a ligand-free form and two in distinct CRF-bound states. The CRFR1 ECD adopts the alpha-beta-betaalpha fold observed for other class B GPCR ECDs, but the N-terminal alpha-helix is significantly shorter and does not contact CRF. CRF adopts a continuous alpha-helix that docks in a hydrophobic surface of the ECD that is distinct from the peptide-binding site of other class B GPCRs, thereby providing a basis for the specificity of ligand recognition between CRFR1 and other class B GPCRs. The binding of CRF is accompanied by clamp-like conformational changes of two loops of the receptor that anchor the CRF C terminus, including the C-terminal amide group. These structural studies provide a molecular framework for understanding peptide binding and specificity by the CRF receptors as well as a template for designing potent and selective CRFR1 antagonists for therapeutic applications.
- Laboratory of Structural Sciences, Van Andel Research Institute, Grand Rapids, Michigan 49503, USA. augie.pioszak@vai.org
Organizational Affiliation: