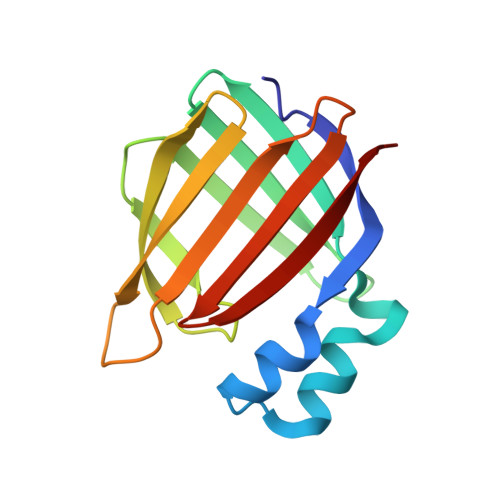
Determining Crystal Structures of Proteins and Protein Complexes by X-Ray Crystallography: X-Ray Crystallographic Studies of the Mutants of Cellular Retinoic Acid Binding Protein Type II Toward Designing a Mimic of Rhodopsin.
Vaezeslami, S., Jia, X., Vasileiou, C., Borhan, B., Geiger, J.H.(null) Thesis