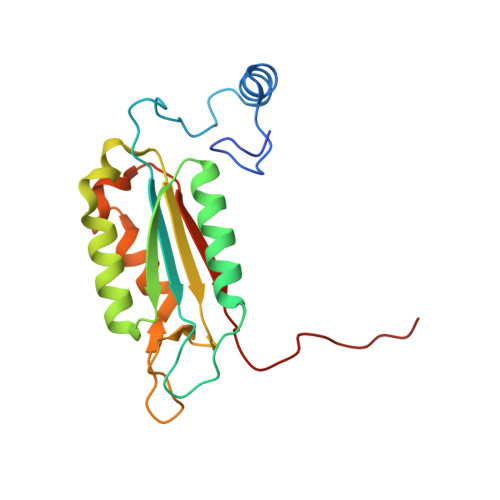
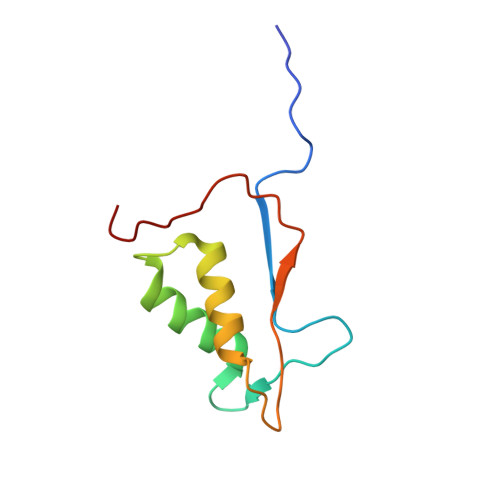
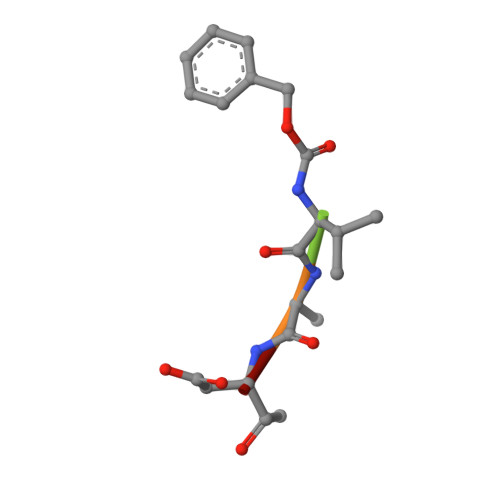
Mutated, structurally altered caspase-1 with decreased enzymatic and increased RIP2-meditated inflammatory activity leads to a new type of periodic fever (ICE fever).
Rosen-Wolff, A., Romanowski, M.J., Ritter, L., Flecks, S., Quoos, N., Gramatt, J., Petzold, C., Nguyen, H.D., Gahr, M., Roesler, J.To be published.