Structure of the DTDP-4-Keto-L-Rhamnose Reductase related protein (other form) from Thermus Thermophilus HB8
Jeyakanthan, J., Kanaujia, S.P., Sekar, K., Satoh, S., Kitamura, Y., Yokoyama, S., Kurmamitsu, S.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Putative uncharacterized protein TTHB138 | 103 | Thermus thermophilus HB8 | Mutation(s): 0 | 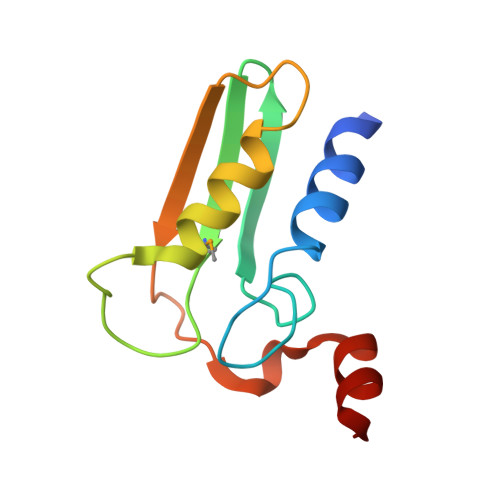 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q53W28 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| P6G Download:Ideal Coordinates CCD File | I [auth A] | HEXAETHYLENE GLYCOL C12 H26 O7 IIRDTKBZINWQAW-UHFFFAOYSA-N |  | ||
| PG4 Download:Ideal Coordinates CCD File | W [auth E] | TETRAETHYLENE GLYCOL C8 H18 O5 UWHCKJMYHZGTIT-UHFFFAOYSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | G [auth A] H [auth A] K [auth B] L [auth B] M [auth B] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| MG Download:Ideal Coordinates CCD File | F [auth A], J [auth B], N [auth C], R [auth D], T [auth E] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A, B, C, D, E | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 175.122 | α = 90 |
| b = 175.122 | β = 90 |
| c = 99.095 | γ = 90 |
| Software Name | Purpose |
|---|---|
| CNS | refinement |
| HKL-2000 | data collection |
| HKL-2000 | data reduction |
| SCALEPACK | data scaling |
| AMoRE | phasing |