Packing Space Expansion of Protein Crystallization Screening with Synthetic Zeolite as a Heteroepitaxic Nucleant
Sugahara, M., Kageyama-Morikawa, Y., Kunishima, N.(2011) Cryst Growth Des 11: 110-120
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
(2011) Cryst Growth Des 11: 110-120
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| xylanase | 190 | Trichoderma longibrachiatum | Mutation(s): 0 EC: 3.2.1.8 | 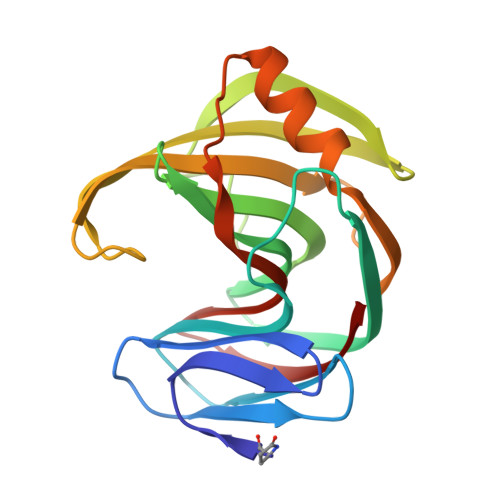 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | F8W669 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| GOL Download:Ideal Coordinates CCD File | B [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| NA Download:Ideal Coordinates CCD File | C [auth A], D [auth A] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| PCA Query on PCA | A | L-PEPTIDE LINKING | C5 H7 N O3 |  | GLN |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 56.755 | α = 90 |
| b = 38.904 | β = 110.48 |
| c = 40.214 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data collection |
| MOLREP | phasing |
| CNS | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |