Structure and Activity of the N-Terminal Substrate Recognition Domains in Proteasomal Atpases.
Djuranovic, S., Hartmann, M.D., Habeck, M., Ursinus, A., Zwickl, P., Martin, J., Lupas, A.N., Zeth, K.(2009) Mol Cell 34: 580
- PubMed: 19481487 Search on PubMed
- DOI: https://doi.org/10.1016/j.molcel.2009.04.030
- Primary Citation Related Structures:
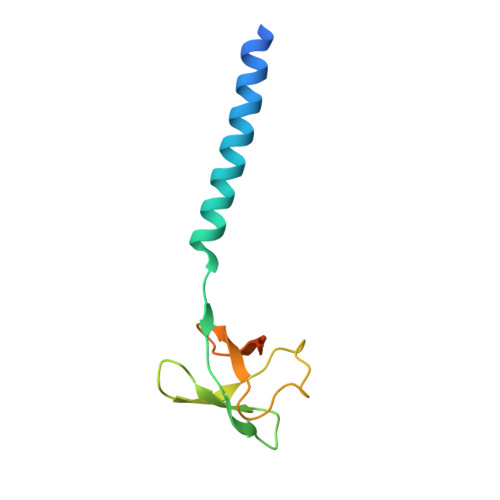
2WFW, 2WG5, 2WG6 - PubMed Abstract:
The proteasome forms the core of the protein quality control system in archaea and eukaryotes and also occurs in one bacterial lineage, the Actinobacteria. Access to its proteolytic compartment is controlled by AAA ATPases, whose N-terminal domains (N domains) are thought to mediate substrate recognition. The N domains of an archaeal proteasomal ATPase, Archaeoglobus fulgidus PAN, and of its actinobacterial homolog, Rhodococcus erythropolis ARC, form hexameric rings, whose subunits consist of an N-terminal coiled coil and a C-terminal OB domain. In ARC-N, the OB domains are duplicated and form separate rings. PAN-N and ARC-N can act as chaperones, preventing the aggregation of heterologous proteins in vitro, and this activity is preserved in various chimeras, even when these include coiled coils and OB domains from unrelated proteins. The structures suggest a molecular mechanism for substrate processing based on concerted radial motions of the coiled coils relative to the OB rings.
- Department of Protein Evolution, Max Planck Institute for Developmental Biology, D-72076 Tübingen, Germany.
Organizational Affiliation: