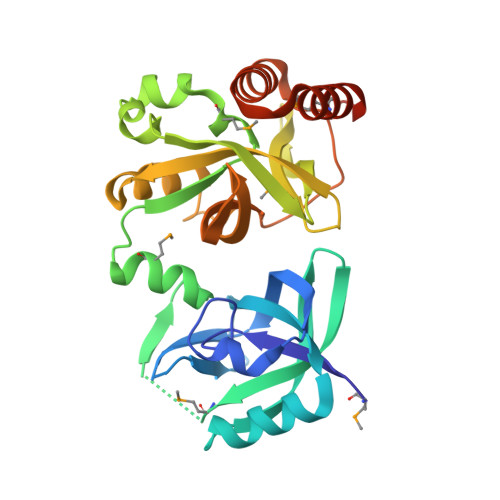
Crystal structure of YkqA from Bacillus subtilis.
Benach, J., Chen, Y., Forouhar, F., Seetharaman, J., Baran, M.C., Cunningham, K., Ma, L.-C., Owens, L., Chen, C.X., Rong, X., Janjua, H., Acton, T.B., Montelione, G.T., Tong, L., Hunt, J.F.To be published.