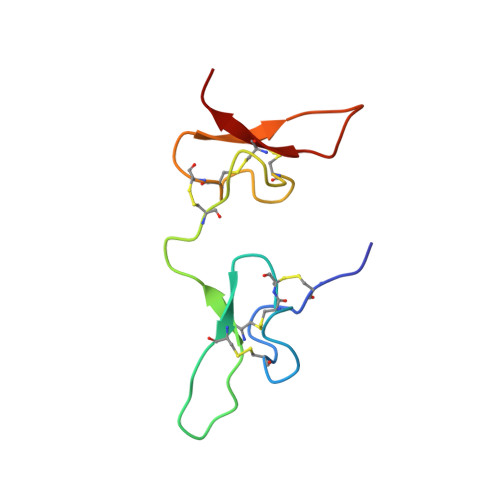
Potent neuroprotection after stroke afforded by a double-knot spider-venom peptide that inhibits acid-sensing ion channel 1a.
Chassagnon, I.R., McCarthy, C.A., Chin, Y.K., Pineda, S.S., Keramidas, A., Mobli, M., Pham, V., De Silva, T.M., Lynch, J.W., Widdop, R.E., Rash, L.D., King, G.F.(2017) Proc Natl Acad Sci U S A 114: 3750-3755
- PubMed: 28320941 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1614728114
- Primary Citation Related Structures:
2N8F - PubMed Abstract:
Stroke is the second-leading cause of death worldwide, yet there are no drugs available to protect the brain from stroke-induced neuronal injury. Acid-sensing ion channel 1a (ASIC1a) is the primary acid sensor in mammalian brain and a key mediator of acidosis-induced neuronal damage following cerebral ischemia. Genetic ablation and selective pharmacologic inhibition of ASIC1a reduces neuronal death following ischemic stroke in rodents. Here, we demonstrate that Hi1a, a disulfide-rich spider venom peptide, is highly neuroprotective in a focal model of ischemic stroke. Nuclear magnetic resonance structural studies reveal that Hi1a comprises two homologous inhibitor cystine knot domains separated by a short, structurally well-defined linker. In contrast with known ASIC1a inhibitors, Hi1a incompletely inhibits ASIC1a activation in a pH-independent and slowly reversible manner. Whole-cell, macropatch, and single-channel electrophysiological recordings indicate that Hi1a binds to and stabilizes the closed state of the channel, thereby impeding the transition into a conducting state. Intracerebroventricular administration to rats of a single small dose of Hi1a (2 ng/kg) up to 8 h after stroke induction by occlusion of the middle cerebral artery markedly reduced infarct size, and this correlated with improved neurological and motor function, as well as with preservation of neuronal architecture. Thus, Hi1a is a powerful pharmacological tool for probing the role of ASIC1a in acid-mediated neuronal injury and various neurological disorders, and a promising lead for the development of therapeutics to protect the brain from ischemic injury.
- Institute for Molecular Bioscience, The University of Queensland, St. Lucia, QLD 4072, Australia.
Organizational Affiliation: