Unfolding of the C-terminal domain of the j-protein zuo1 releases autoinhibition and activates pdr1-dependent transcription.
Ducett, J.K., Peterson, F.C., Hoover, L.A., Prunuske, A.J., Volkman, B.F., Craig, E.A.(2013) J Mol Biology 425: 19-31
- PubMed: 23036859 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2012.09.020
- Primary Citation Related Structures:
2LWX - PubMed Abstract:
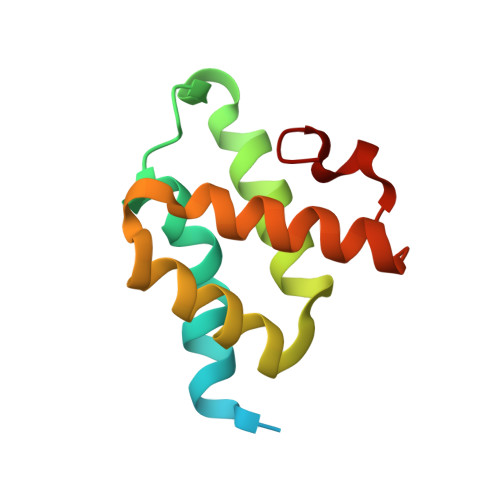
The C-terminal 69 residues of the J-protein Zuo1 are sufficient to activate Pdr1, a transcription factor involved in both pleiotropic drug resistance and growth control. Little is understood about the pathway of activation by this primarily ribosome associated Hsp40 co-chaperone. Here, we report that only the C-terminal 13 residues of Zuo1 are required for activation of Pdr1, with hydrophobic residues being critical for activity. Two-hybrid interaction experiments suggest that the interaction between this 13-residue Zuo1 peptide and Pdr1 is direct, analogous to the activation of Pdr1 by xenobiotics. However, simply dissociation of Zuo1 from the ribosome is not sufficient for induction of Pdr1 transcriptional activity, as the C-terminal 86 residues of Zuo1 fold into an autoinhibitory left-handed four-helix bundle. Hydrophobic residues critical for interaction with Pdr1 are sequestered within the structure of this C-terminal domain (CTD), necessitating unfolding for activation. Thus, although expression of the CTD does not result in activation, alterations that destabilize the structure cause induction of pleiotropic drug resistance. These destabilizing alterations also result in dissociation of the full-length protein from the ribosome. Thus, our results are consistent with an activation pathway in which unfolding of Zuo1's C-terminal helical bundle domain results in ribosome dissociation followed by activation of Pdr1 via a direct interaction.
- Department of Biochemistry, University of Wisconsin, Madison, WI 53706, USA.
Organizational Affiliation: